Volume 43 Number 4
Examining the association of immunosuppressants and wound healing: a narrative review
Aria Appoo, Brandon L Christensen, Ranjani Somayaji
Keywords wound healing, Calcineurin inhibitors, glucocorticoids, immunosuppressants, monoclonal antibodies, mTOR inhibitors
For referencing Appoo A, Christensen BL and Somayaji R . Examining the association of immunosuppressants and wound healing: a narrative review. WCET® Journal 2023;43(4):32-39.
DOI
10.33235/wcet.43.4.32-39
Submitted 1 February 2023
Accepted 23 May 2023
Abstract
Objective A review of recent literature to provide clinicians with an understanding of how different classes of immunosuppressants affect wound healing.
Data sources A literature search was conducted in PubMed, Google Scholar, and the University of Calgary Health Sciences Library.
Study Selection Studies chosen for inclusion were screened initially based on title using key words including “immunosuppressive medication, wound healing, and immunosuppression.” If the title and/or abstract contained these key words and addressed wound healing related to immunosuppressant medications and had been published after 2000, they were included in the review. When human data was not available for an immunosuppressant (class), animal studies were included.
Data Extraction Selected papers underwent full text review and summarisation.
Data Synthesis Data were synthesised in a descriptive manner. Corticosteroids and mechanistic target of rapamycin (mTOR) inhibitors most consistently demonstrate detrimental effects on wound healing. For other classes of immunosuppressants, evidence is limited with varying effects on wound healing described.
Conclusions Larger high-quality studies are required to better understand the effects of immunosuppressants particularly with development of new classes of these drugs on wound healing in order to identify those at highest risk of impairing wound healing.
Introduction
Immunosuppressants are medications with a variety of indications including in solid organ and hematopoietic transplants along with autoimmune diseases. They function by suppressing the activity of various components of the adaptive immune system thus diminishing the cascade of inflammatory response to normal host tissue or modulating the natural rejection response to transplanted materials.1 The main classes of immunosuppressants are corticosteroids/glucocorticoids,2,3 calcineurin inhibitors,2,4,5 mechanistic target of rapamycin (mTOR) inhibitors,2,4 polyclonal antibodies,2,4 monoclonal antibodies,2,4 and antiproliferative agents.2 For the purpose of this review, wounds are defined as an opening in the skin as a result of surgery, trauma, or disease that is susceptible to infection.
The immune system plays an important role in the prevention of infection but also the healing process of wounds, with the inflammatory effects leading to cellular proliferation and secretion of important intra and extracellular components.6 With immunosuppressants, the immune system is modulated, thus potentially affecting a wound’s healing time and susceptibility to infection.7 With a growing number of patients on immunosuppressing medications, particularly post-surgical transplant patients, the effect of immunosuppressants on wound healing is an important issue to be considered. This review article aims to provide clinicians with an understanding of how different classes of immunosuppressants affect wound healing.
Materials/methods
A literature search between 2000 through 2021 was conducted using the generic names of a number of commonly used immunosuppressants (glucocorticoids/corticosteroids, mTOR inhibitors, methotrexate, monoclonal antibodies, polyclonal antibodies, calcineurin inhibitors, mycophenolate, azathioprine) as well as the terms “wound healing” and “immunosuppression”. The primary database searched was PubMed. This was supplemented by Google Scholar and the University of Calgary Health Science Library database. When possible, the search was in the following format “immunosuppressant name [MeSH Terms] AND “wound healing [MeSH Terms].” If the immunosuppressant name was not available as a MeSH term, then the term was searched with no restriction applied. Article titles were then screened for relevance to the review based on whether wound healing was described in relation to the immunosuppressive medication/class. The abstract was screened similarly and included in the review accordingly. If the article compared various immunosuppressants, discussed their effects on wound healing, measured wound healing or deleterious effects on wounds it was considered relevant. If search terms did not identify human subject studies, then studies that used animals to evaluate immunosuppressive effects on wound healing were included. When no data was available from 2000 onwards, a historic search was conducted for the relevant immunosuppressive medications.
All included studies were summarised descriptively including immunosuppressive mechanism of action, study subjects, and evidence of effects on wound healing.
Results
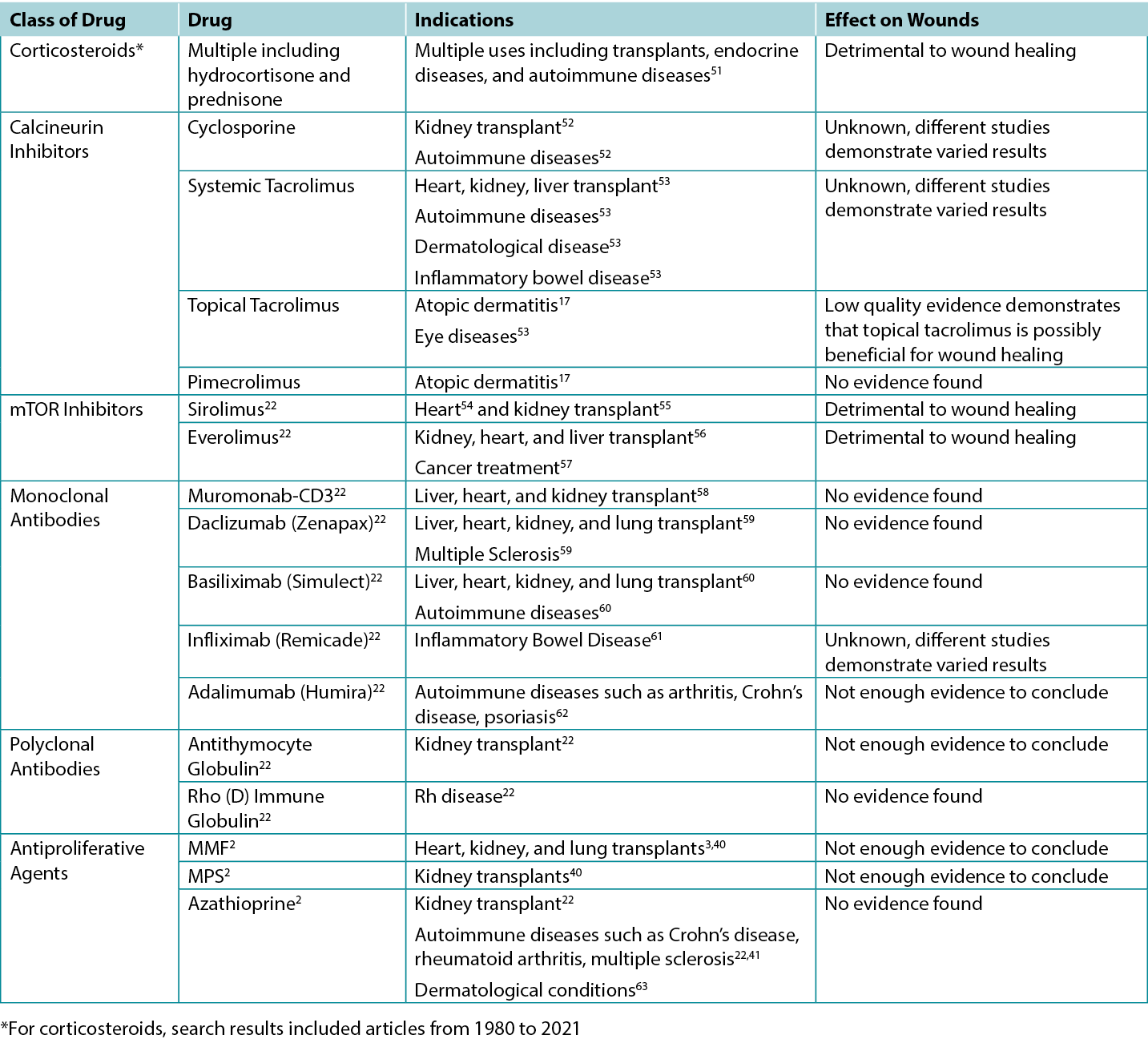
The studies screened relevant to this review were used to inform the various categories outlined below. A total of 200 article titles and abstracts were initially screened and 61 articles were included for review. Summary tables highlighting the results of clinical and animal studies are summarised in Table 1. The specific indications for various immunosuppressants including their possible impacts on wounds are outlined in Table 2.
Table 1. Studies Comparing the Effects of Different Immunosuppressants on Wounds
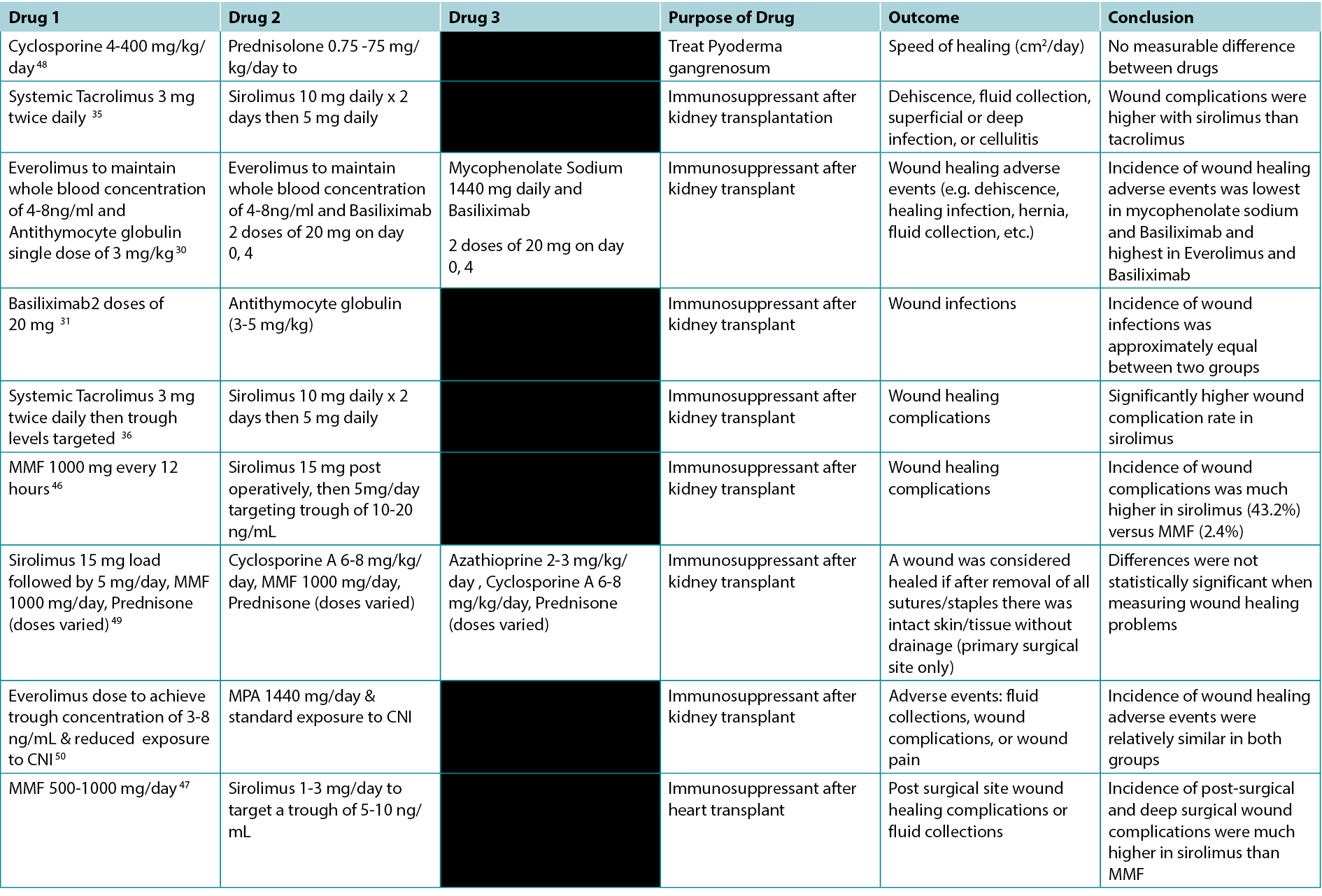
Table 2. Overview of Indications and Wound Effects of Various Immunosuppressants
Calcineurin Inhibitors
Calcineurin inhibitors (CNI) are used as immunosuppressants for a variety of different autoimmune diseases, organ transplants, dermatological conditions, and in chronic wounds.8 There are three main types of CNI’s: cyclosporine (systemic), tacrolimus (systemic and topical), pimecrolimus (topical).9 CNI’s work by binding to part of the calcineurin molecule found in human cells, thus stopping the release of certain cytokines that are responsible for activating T cells.8 Therefore, CNI’s disable one of the main arms of the body’s adaptive immune response.
Systemic
There are a lack of studies directly focusing on the effects of CNI’s on wound healing in humans; however, many basic science studies on animals have been performed. Two such studies using rats compared the effect of various doses of systemic tacrolimus versus a control, testing the breaking strengths of the wounds created through surgery. One of the rat studies concluded that tacrolimus does not affect wound healing10 while the other study concluded that tacrolimus is detrimental to wound healing.11 At the same time, case reports using systemic tacrolimus as a treatment for ulcers in a person with lichen planus and pyoderma gangrenosum demonstrated treatment success with this therapy.12
No recent human studies were found regarding the effect of cyclosporine and wound healing. Two other studies using rats also yielded contradictory results. These rat studies focused on the effects of cyclosporine on different markers in the body that signify effective wound healing. One study comparing cyclosporine to methylprednisone demonstrated that cyclosporine had no suppressive effect on the various inflammatory and biochemical markers compared to the glucocorticoid therapy13 whereas the other study showed that cyclosporine had a negative effect on the markers.14 In a different study, after receiving a lung transplant, dogs were assigned to a specific immunosuppressant drug or no immunosuppressant drug and, similar to control, Cyclosporine A was shown to have no significant effect on the healing of the surgical wound as measured by breaking strength in comparison to glucocorticoid and azathioprine immunosuppression.15 Overall, the literature on systemic calcineurin inhibitors and wound healing is dated with a heterogeneity of comparators and mixed results on wound healing.
Topical
Calcineurin inhibitors (tacrolimus and pimecrolimus) are used as topical ointments most commonly for dermatological conditions such as atopic dermatitis but also for chronic dermatologic conditions such as pyoderma gangrenosum.16–18 Some case studies have shown tacrolimus effective at healing complex leg ulcers in the context of venous insufficiency or necrobiosis lipoidica, when regular treatment strategies have been ineffective.19,20 Furthermore, a rat-based study with acute cutaneous injury demonstrated that wounds treated with topical tacrolimus healed equally as quickly as the control (petrolatum).21
Monoclonal Antibodies:
A variety of different monoclonal antibodies (mAb) therapies exist with indications in transplants and autoimmune disorders such as rheumatoid arthritis and psoriasis.22 In general, mAb work by binding to different receptors and antigens to inhibit the effect of cytokines and other signal pathways that activate the immune system.22 A small prospective cohort study among patients with rheumatoid arthritis undergoing orthopedic surgery demonstrated that there was no increased risk of surgical wound infections or healing complications in patients on infliximab compared to conventional therapy.23 Furthermore, a case report claims that topical infliximab was helpful in healing leg ulcers that were resistant to standard treatment suggesting that inhibiting tumor necrosis factor alpha is helpful to wound healing.24 Similar findings were concluded in a case report for treatment of pyoderma gangrenosum with infliximab resulting in improvement of the ulcer.25 Inhibiting tumor necrosis factor alpha was shown to be further associated with wound healing in venous leg ulcers through the use of Adalimumab systemically.26 However, another study used infliximab on rat abdominal wounds and demonstrated that the tensile strength was significantly lower in the wounds of rats who had been given infliximab compared to those who were the control.27 Overall, the limited literature on the subject to date suggests clinical outcomes thus far are favorable in terms of wound healing.
Polyclonal Antibodies
Polyclonal antibodies (pAb’s) are very similar to mAb’s in function, with slightly varied mechanism of action.28,29 Unlike mAb’s, a group of polyclonal antibodies are created from many different lines of B cells and within the group different pAb’s bind to different epitopes of an antigen. In contrast, mAb’s come from a single line of B cells and can bind to only one antigen.28,29 There are two main pAb’s: Antithymocyte Globulin and Rho (D) Immune Globulin.22 Antithymocyte Globulin also known as Antihuman Thymocyte Globulin is most commonly used as an immunosuppressant after kidney transplantation and works by binding to a variety of lymphocytes and depleting the number of T cells in the body.22 Rho (D) Immune Globulin is used in pregnancies where the mother is Rh- and the fetus is Rh+ and essentially stops the formation of anti Rh+ antibodies in the mother.22
Few studies looking at the effects of pAb’s on wound healing were found. Two studies outlined the effects of antithymocyte globulin and Basiliximab. The first study in patients with renal transplants, where both drugs were combined with everolimus, showed higher rates of adverse effects of wound healing in Basiliximab. 30 The second study demonstrating that the incidence of wound infections was equal in patients taking Basiliximab versus antithymocyte globulin post renal transplant.31 Since white blood cells play a key role in wound healing through secreting necessary cytokines and preventing infection,32 it would be reasonable to hypothesise that antithymocyte globulin will affect wound healing since it reduces the number of white blood cells as well as their regulatory mechanisms.
mTOR Inhibitors
Mechanistic target of rapamycin (mTOR) inhibitors are immunosuppressive drugs that works by interacting with proteins in complex signalling pathways to prevent cells moving into the S phase of the cell cycle and therefore suppressing proliferation.3,22 mTOR predominantly targets T cells, but can also affect B cells.3 Interestingly, mTOR can cause an increase of production of certain inflammatory cytokines such as interleukin-6 and decrease production of interleukin-10, an anti-inflammatory cytokine.3 There are two main mTOR inhibitors: sirolimus and everolimus.3,22 In general, mTOR inhibitors have a variety of applications including cancer therapy.3,22 As an immunosuppressant, they are used after transplants.3,22 Everolimus has been shown to inhibit the proliferation of fibroblast in in vitro models33 suggesting that everolimus could have negative consequences for wound healing since fibroblasts are essential for creating an extracellular matrix and creating a frame for other cells.34 In a study comparing sirolimus and systemic tacrolimus, sirolimus had a wound complication rate of 47% compared to tacrolimus with only 8%.35 This is consistent with another study by Larson et al demonstrating higher wound complications with sirolimus compared to tacrolimus.36 Interestingly, obese patients on sirolimus had very high rates of wound complications leading to the study excluding all patients with obesity.36 In line with previous findings, a study on rats showed that increased sirolimus doses decreases wound strength, which the authors hypothesising this effect may be due to lower levels of VEGF and nitric oxide in rats receiving higher doses of sirolimus.37 In one review authors concluded that mTOR inhibitors are harmful to wound healing in high doses, but seem to have a neutral effect in low does.38 Given early concerns with mTOR inhibitors and wound healing, regimens using these immunosuppressive agents have evolved with lower doses of the mTOR inhibitors and combination therapy. In the large TRANSFORM randomised control trial investigators compared everolimus plus reduced dose CNI to mycophenolic acid plus standard dose CNI (standard care) in patients with renal transplants and showed wound related adverse events were not statistically different (20.6% vs 17.3%; RR 1.19 95%CI 0.99 to 1.43).39 One limitation of this study was close monitor of everolimus concentrations and difficulties achieving the targeted plasma concentrations C0 between 3 and 8 ng/mL.39 Overall, review of the current literature is suggestive that mTOR inhibitors have a detrimental effect on wound healing, especially at higher doses, and that improved dosing regimens may lessen or mitigate this risk.
Antiproliferative Agents
There are three regularly used antiproliferative agents: Mycophenolate mofetil (MMF) and Mycophenolate sodium (MPS) (both inosine monophosphate dehydrogenase (IMPDH) inhibitors) and azathioprine.2,40 IMPDH inhibitors have a similar effect to mTOR inhibitors in terms of their mechanism of immunosuppression. In the body, MMF and MPS are converted into mycophenolic acid which blocks a portion of a pathway that is crucial for DNA synthesis, to decrease proliferation of T cells and B cells.2,5,40 MMF is used for its immunosuppressive effect in heart, kidney, and lung transplants.3,40 MPS is used for kidney transplants.40 Azathioprine is used as an immunosuppressive drug for kidney transplants and autoimmune diseases including rheumatoid arthritis, Crohn’s disease, and multiple sclerosis.22,41 In the body, azathioprine reacts with glutathione and is converted into 6-mercaptupurine after which additional metabolites are generated ultimately blocking purine synthesis and stimulation of T-cells.3,22
When a study compared two different doses of MMF in kidney transplant recipients, incidence of wounds requiring surgical intervention were not significantly different, similarly for wounds treated with local wound care.42 Based on review of article titles as part of our search, no studies regarding the sole effect of azathioprine on external wound healing in humans were found. When azathioprine was compared to a placebo in a rat study, the wounds of the rats who were on azathioprine took longer to heal than those on the placebo suggesting that azathioprine can have detrimental effects on wound healing, but the extent that it would affect humans is unclear.43
Anti-metabolite
Methotrexate is a commonly used folate antagonist immunosuppressive agent, with indications in many rheumatologic disorders. It also has antineoplastic activity in higher doses. Upon absorption, it enters the cell and is converted to methotrexate polyglutamates where it competes for dihydrofolate reductase thus preventing the transformation of folic acid for its use in the building of nucleic acids.6 Like other agents described, there is experimental animal studies with in vitro studies suggesting impairment in wound healing but these effects have not been borne out in clinical studies, particularly in post-surgical wounds.6 Thus, it is recommended this drug be continued postoperatively.
Corticosteroids/Glucocorticoids
Glucocorticoids prevent the formation of inflammatory chemicals like cytokines, cell adhesion molecules, and complement factors.3 By inhibiting interleukin-2 formation, glucocorticoids also prevent T cell proliferation and activation.22 They also impair monocytes and B cells.3,22 Glucocorticoids were the first antirejection drug created, but as time has passed, there has been a movement to phase them out due to their serious side effects.3,22 There is consensus that glucocorticoids are highly detrimental to wound healing since they interfere with many key stages of wound healing such as collagen deposition and synthesis, angiogenesis, fibroblast proliferation, growth factors, and phagocytosis among others.2,3,44,45
Practical considerations for health care providers
Persons with compromised immune systems (due to medications, co-morbidities or age extremes) require additional considerations for chronic wound management. Specific to immunosuppressive medications, health care providers should take a careful history not only of the medications and dosing (including changes in dosing), but also of the underlying conditions requiring these medications (e.g. autoimmune disorders, organ transplantation). As many immunosuppressive medications can impair wound healing, it is crucial for health care providers to assess healing potential early on to set and manage patient expectations. Early referral to medical or surgical specialists to assist with wound care and a team-based approach will be essential given the increased complexity of these individuals. In some cases where wounds are not healing, alternate goals of care for the wound may need to be set (e.g. maintenance or non-healable) if immunosuppressive doses cannot be reduced (assuming it is contributing to poor healing) and should be done in consultation with their primary or specialist care providers. As individuals and populations with comorbidities live longer, taking care of persons with chronic wounds on immunosuppressive medications will become increasingly common and must be recognised early by the wound care clinicians.
Discussion
With the ongoing advances in medicine, the need for immunosuppression in the context of transplant, autoimmune disease and malignancy has increased. In our review, we highlight the poverty of robust studies in this field and highlighted the mixed effects of various immunosuppression on wound healing. High quality evidence exists with respect to the deleterious effects of glucocorticoid therapy as well as mTOR therapy, particularly sirolimus on wound healing. Furthermore, when 4 studies compared sirolimus to either MMF or systemic tacrolimus, all the studies demonstrated sirolimus to be associated with an increased incidence of wound complications.35,36,46,47
The literature on agents such as systemic CNIs is mixed with some suggesting adverse effects on wounds and others suggesting benefit, necessitating additional study focusing directly on this question. Newer topical CNIs have shown little impact on delayed wound healing and in some cases benefit, but additional investigation is warranted for their use in chronic wounds directly. Reassuringly, antiproliferative agents, antimetabolite, and newer monoclonal antibiotics have not shown signal toward diminished healing. However, additional study is needed given the poverty of evidence on wound healing in mAB therapy. Overall, our review found the evidence in this area is dated with variable conclusions surrounding the effects of the immunosuppressant on wound healing. In addition, we found little evidence using human subjects. In general, when immunosuppressives are prescribed after transplants to prevent rejection, patients take more than one drug to effectively prevent rejection. Therefore, challenges exist in performing human studies evaluating the effects of individual drugs in isolation.
As we did not conduct a systematic review, certain articles may have been excluded. We did identify a few key reviews before conducting the search and ensured they were present in the search as one form of validation. As well, given the paucity of literature in this area particularly as it relates to the wound care field, a narrative review adds value to educate and increase awareness when working with individuals on these medications.
With the growing need for immunosuppression, additional study in this field is critical. Future directions include conducting more studies in animal subjects with newer classes of immunosuppressants to identify potential pathways to delayed wound healing and potential ways to mitigate such effects. In addition, more high-quality studies are required to evaluate both individual and combination immunotherapies to better understand the risks and how different immunosuppressants may impact wound healing. In order to explore immunosuppressants as a potential treatment for chronic or complex wounds, it is important for future studies to be conducted on a larger scale, control for confounding clinical factors, such as through randomised control trials.
Conclusions
In conclusion, immunosuppressants range from possibly beneficial to floridly deleterious in wound healing. While there is little conclusive evidence in this field, the effects of immunosuppressants on wound healing is worth exploring to better tailor immunosuppression to patients at risk for or experiencing chronic non-healing wounds. Reassuringly, our findings suggest not all immunosuppressants are harmful with some potentially offering benefit to wound treatment when conventional therapies has failed, opening up the possibility of a new treatment option for wounds.
Acknowledgements
University of Alberta Highschool Youth Researcher Summer Program.
Conflict of Interest
The authors declare no conflicts of interest.
Funding
The authors received no funding for this study.
考察免疫抑制剂与伤口愈合的相关性:叙述性综述
Aria Appoo, Brandon L Christensen, Ranjani Somayaji
DOI: 10.33235/wcet.43.4.32-39
摘要
目的 对近期文献进行综述,促进临床医了解不同类别的免疫抑制剂如何影响伤口愈合。
数据来源 在PubMed、Google Scholar和卡尔加里大学健康科学图书馆进行了文献检索。
研究选择 根据标题,使用Åg免疫抑制药物、伤口愈合和免疫抑制Åh等关键词对选择纳入的研究进行了初步筛选。如果标题和/或摘要中包含这些关键词,并阐述了与免疫抑制剂药物相关的伤口愈合,且在2000年之后发表,则将其纳入综述。无法获得免疫抑制剂(类)的人体数据时,则纳入动物研究。
数据提取 对选定的论文进行全文审阅和总结。
数据合成 以描述性方式合成数据。皮质类固醇和雷帕霉素(mTOR)抑制剂机制性靶标最常对伤口愈合产生不利影响。对于其他类别的免疫抑制剂,证据有限,对伤口愈合的影响也不尽相同。
结论 为了更好地了解免疫抑制剂对伤口愈合的影响,尤其是随着新类别药物的开发,需要进行更大规模的高质量研究,以确定最有可能损害伤口愈合的药物类别。
引言
免疫抑制剂是一类具有多种适应症的药物,包括实体器官和造血干细胞移植以及自身免疫性疾病。这类药物通过抑制适应性免疫系统各种组分的活性发挥作用,进而减少对正常宿主组织的级联炎症反应或调节对移植材料的自然排斥反应1。免疫抑制剂的主要类别包括皮质类固醇/糖皮质激素2,3、钙调磷酸酶抑制剂2,4,5、雷帕霉素(mTOR)抑制剂机制性靶标2,4、多克隆抗体2,4、单克隆抗体2,4和抗増殖剂2。考虑到本综述的目的,伤口定义为手术、外伤或疾病导致的易感染皮肤开口。
免疫系统在预防感染以及伤口愈合过程中发挥重要作用,炎症效应会导致细胞増殖并分泌重要的细胞内和细胞外组分6。使用免疫抑制剂可以调节免疫系统,从而可能影响伤口的愈合时间和对感染的易感性7。随着越来越多的患者使用免疫抑制药物,尤其是术后移植患者,免疫抑制剂对伤口愈合的影响成为一个需考虑的重要问题。本综述文章旨在促进临床医生了解不同类别的免疫抑制剂对伤口愈合的影响。
材料/方法
使用多种常用免疫抑制剂(糖皮质激素/皮质类固醇、mTOR抑制剂、甲氨蝶呤、单克隆抗体、多克隆抗体、钙调磷酸酶抑制剂、麦考酚酯、硫唑嘌呤)的通用名以及Åg伤口愈合Åh和Åg免疫抑制Åh这两个术语,对2000年至2021年间的文献进行了检索。检索的主要数据库为PubMed。此外,还检索了Google Scholar和卡尔加里大学健康科学图书馆数据库以进行补充。在可能的情况下,使用以下格式进行检索:Åg免疫抑制剂名称[MeSH术语]和Åg伤口愈合[MeSH术语]Åh。如果无法获得免疫抑制剂名称作为MeSH术语,则检索该术语,不加任何限制。然后,根据是否描述了伤口愈合与免疫抑制药物/类别的关系,筛选了与综述相关的文章标题。采用类似方法对摘要进行了筛选,并相应纳入综述。如果文章对各种免疫抑制剂进行了比较,讨论了这些免疫抑制剂对伤口愈合的影响,并测量了伤口愈合或对伤口的不利影响,则认为该文章具有相关性。如果使用检索词未发现人类受试者研究,则纳入使用动物评价免疫抑制对伤口愈合影响的研究。如果无法获得2000年之后的数据,则对相关免疫抑制药物进行历史检索。
对所有纳入的研究进行了描述性总结,包括免疫抑制作用机制、研究受试者和对伤口愈合影响的证据。
结果
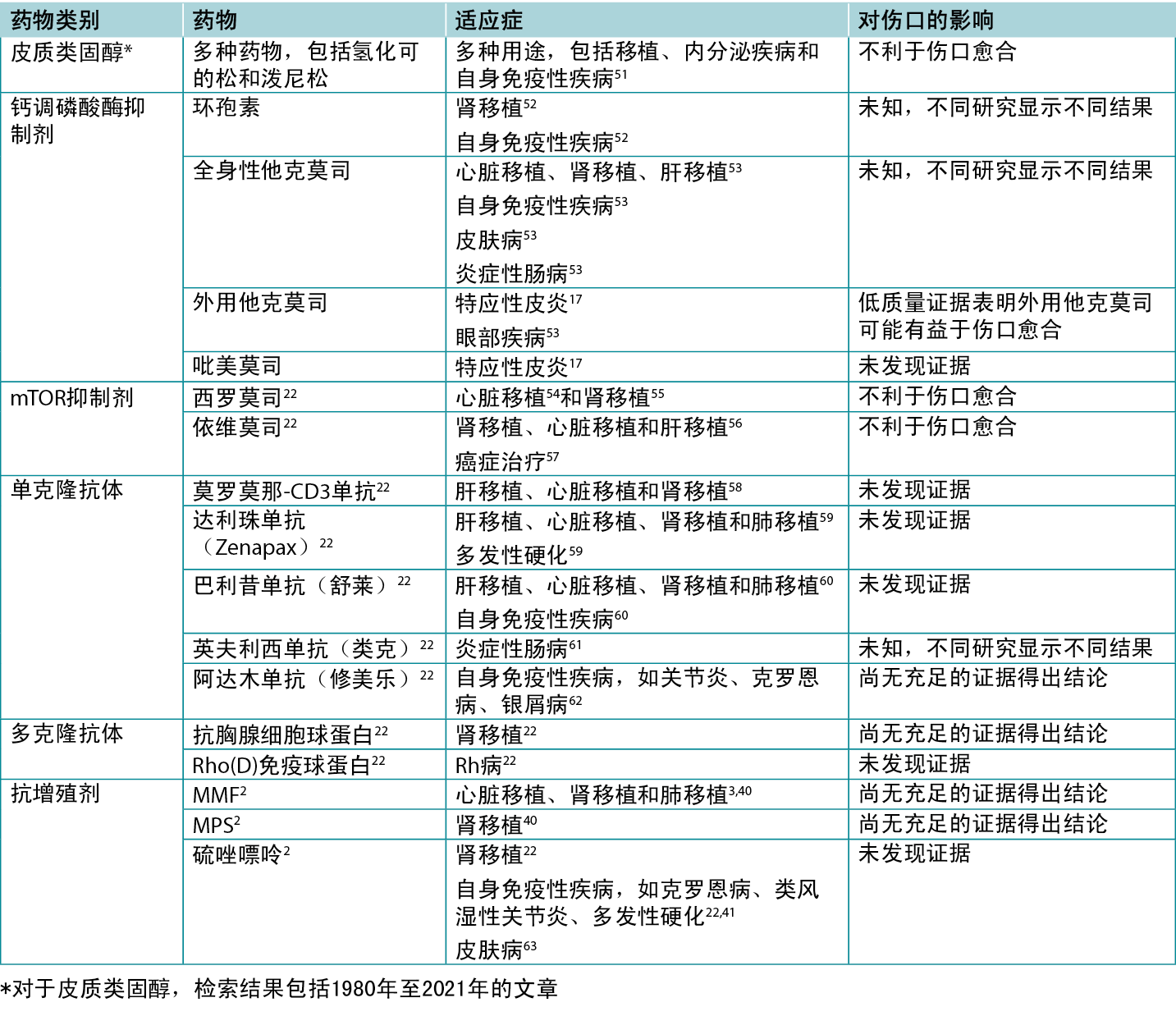
筛选出的与本综述相关的研究用于提供下文所述各类别的相关信息。初步筛选了共200篇文献的标题和摘要,并纳入了61篇文献进行综述。临床研究和动物研究结果的汇总表总结见表1。各种免疫抑制剂的特定适应症(包括其对伤口的可能影响)的概述见表2。
表1.比较不同免疫抑制剂对伤口影响的研究
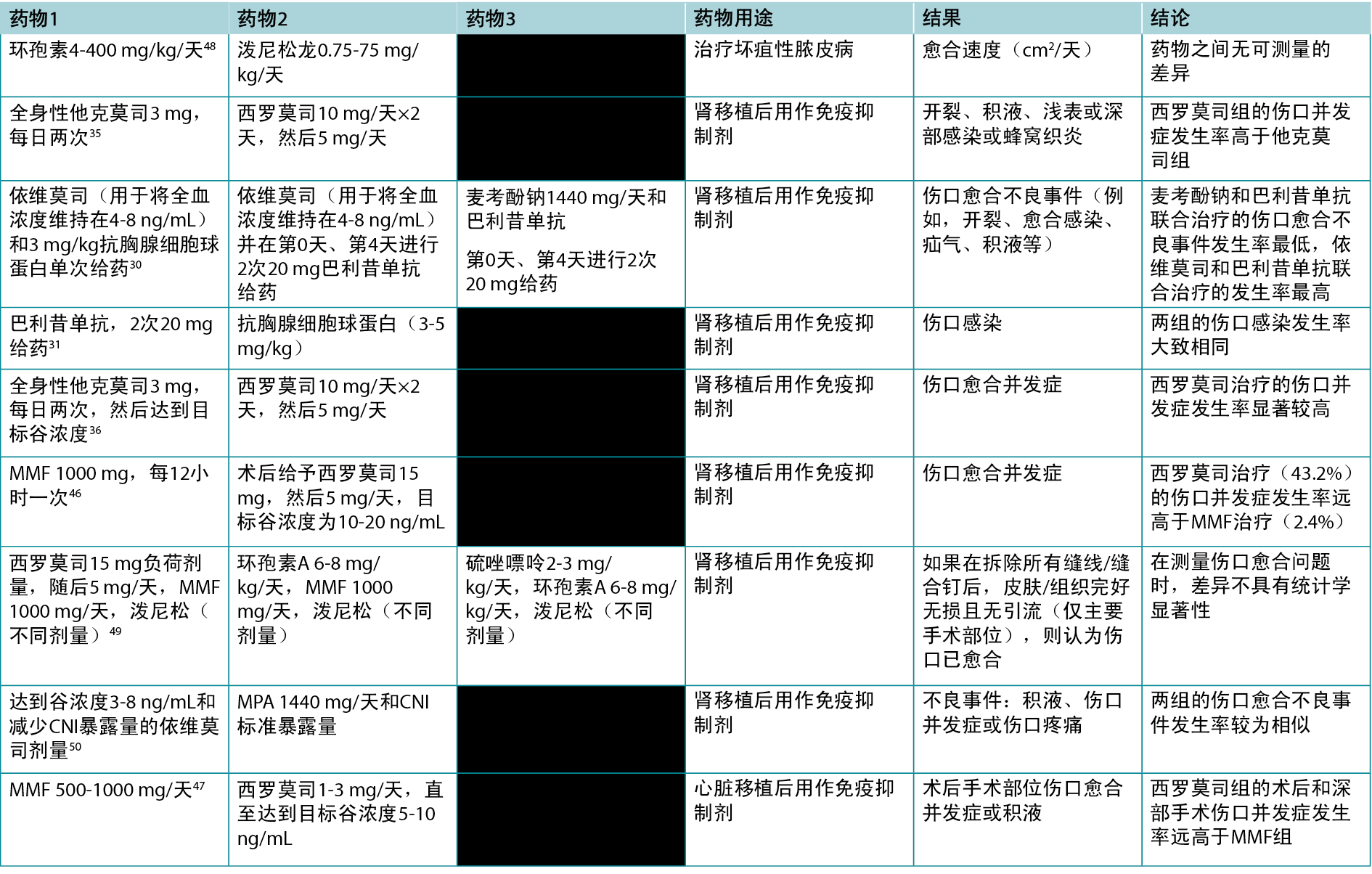
表2.各种免疫抑制剂的适应症和伤口影响概述
钙调磷酸酶抑制剂
钙调磷酸酶抑制剂(CNI)是一种免疫抑制剂,可用于治疗各种不同的自身免疫性疾病、器官移植、皮肤病和慢性伤口8。CNI主要有三种类型:环孢素(全身用)、他克莫司(全身用和外用)、吡美莫司(外用)9。CNI通过与人体细胞中的部分钙调磷酸酶分子结合发挥作用,从而阻止负责激活T细胞的某些细胞因子的释放8。因此,CNI干扰了人体适应性免疫应答的一个主要分支。
全身
缺乏直接关注CNI对人体伤口愈合影响的研究;但是,已对动物进行了许多基础科学研究。使用大鼠进行的两项此类研究比较了不同剂量的全身性他克莫司与对照药物的作用,检测了手术伤口的断裂强度。其中一项大鼠研究得出结论,认为他克莫司不会影响伤口愈合10,而另一项研究则得出结论,认为他克莫司会对伤口愈合产生不利影响11。同时,使用全身性他克莫司治疗扁平苔藓和坏疽性脓皮病患者溃疡的病例报告证实了该治疗方法取得成功12。
未发现关于环孢素和伤口愈合影响的最新人体研究。另外两项大鼠研究也得出了相互矛盾的结果。这些大鼠研究重点关注环孢素对体内不同标志物的影响,这些标志物表示有效的伤口愈合。一项比较环孢素与甲泼尼龙的研究表明,与糖皮质激素治疗相比,环孢素对各种炎症和生化标志物没有抑制作用13,而另一项研究表明,环孢素对标志物有负面影响14。在一项不同的研究中,犬在接受肺移植手术后被分配至特定的免疫抑制剂药物组或无免疫抑制剂药物组,研究结果显示,与对照组相似,环孢素A与糖皮质激素和硫唑嘌呤免疫抑制剂相比,对手术伤口的愈合无显著影响(以断裂强度为衡量指标)15。总体而言,关于全身性钙调磷酸酶抑制剂和伤口愈合的文献年代久远,对照药物不尽相同,对伤口愈合影响的结果也不同。
外用
钙调磷酸酶抑制剂(他克莫司和吡美莫司)作为外用软膏最常用于治疗特应性皮炎等皮肤病,同时也可用于治疗坏疽性脓皮病等慢性皮肤病16-18。一些病例研究表明,当常规治疗策略无效时,他克莫司可有效愈合静脉功能不全或类脂质渐进性坏死背景下的复杂腿部溃疡19,20。此外,一项大鼠急性皮肤损伤研究表明,外用他克莫司治疗伤口的愈合速度与对照药物(凡士林)相同21。
单克隆抗体:
目前存在多种不同的单克隆抗体(mAb)治疗,适用于移植和自身免疫性疾病,如类风湿性关节炎和银屑病22。一般而言,mAb通过与不同的受体和抗原结合来抑制激活免疫系统的细胞因子和其他信号通路的作用22。在接受骨科手术的类风湿性关节炎患者中进行的一项小型前瞻性队列研究表明,与常规治疗相比,接受英夫利昔单抗治疗的患者发生手术伤口感染或愈合并发症的风险并未増加23。此外,一份病例报告称,外用英夫利昔单抗有助于促进对标准治疗产生耐药性的下肢溃疡愈合,表明抑制肿瘤坏死因子Éø有助于伤口愈合24。在使用英夫利昔单抗治疗坏疽性脓皮病来改善溃疡的病例报告中得出了类似的结果25。通过使用全身性阿达木单抗,证明抑制肿瘤坏死因子Éø进一步与下肢静脉性溃疡的伤口愈合相关26。然而,另一项研究使用英夫利西单抗治疗大鼠腹部伤口,结果表明,接受英夫利西单抗治疗的大鼠伤口的抗张强度显著低于对照组27。总体而言,迄今为止关于该主题的有限文献表明,伤口愈合方面的临床结局是有利的。
多克隆抗体
多克隆抗体(pAb)的作用与mAb非常相似,作用机制略有不同28,29。与mAb不同,一组多克隆抗体由多种不同的B细胞系产生,在这组多克隆抗体中,不同的pAb与抗原的不同表位结合。相比之下,mAb来自单个B细胞系,只能与一种抗原结合28,29。存在两种主要的pAb:抗胸腺细胞球蛋白和Rho(D)免疫球蛋白22。抗胸腺细胞球蛋白也称为抗人胸腺细胞球蛋白,最常用于肾移植后的免疫抑制剂,通过与多种淋巴细胞结合并消耗体内T细胞的数量发挥作用22。Rho(D)免疫球蛋白用于母亲为Rh-且胎儿为Rh+的妊娠,基本可停止母亲体内抗Rh+抗体的形成22。
观察pAb对伤口愈合影响的研究较少。两项研究概述了抗胸腺细胞球蛋白和巴利昔单抗的作用。在肾移植患者中进行的第一项研究中,两种药物均与依维莫司联合使用,结果显示巴利昔单抗的伤口愈合不良反应发生率更高30。第二项研究表明,肾移植后使用巴利昔单抗和抗胸腺细胞球蛋白的患者伤口感染的发生率相同31。白细胞可分泌必要的细胞因子并预防感染,在伤口愈合中发挥关键作用32,因此可合理假设抗胸腺细胞球蛋白会影响伤口愈合,原因在于其会减少白细胞数量及其调节机制。
mTOR抑制剂
雷帕霉素(mTOR)抑制剂机制性靶标是一类免疫抑制药物,通过与复杂信号通路中的蛋白相互作用,阻止细胞进入细胞周期的S期,从而抑制细胞増殖3,22。mTOR主要靶向T细胞,但也可能影响B细胞3。值得注意的是,mTOR可増加白细胞介素-6等炎性细胞因子的生成并减少抗炎细胞因子白细胞介素-10的生成3。mTOR抑制剂主要有两种:西罗莫司和依维莫司3,22。一般而言,mTOR抑制剂有多种用途,包括癌症治
疗3,22。作为免疫抑制剂,可在移植后使用这两种药物3,22。经证明,依维莫司在体外模型中可抑制成纤维细胞的増殖33,表明依维莫司可能会对伤口愈合产生负面影响,因为成纤维细胞对于生成细胞外基质和为其他细胞创建框架至关重要34。在一项比较西罗莫司和全身性他克莫司的研究中,西罗莫司的伤口并发症发生率为
47%,而与他克莫司仅为8%35。这与Larson等人进行的另一项研究结果一致,即西罗莫司的伤口并发症发生率高于他克莫司36。值得注意的是,接受西罗莫司的肥胖患者的伤口并发症发生率具有非常高,因此该研究排除了所有肥胖患者36。与先前的研究结果一致,一项大鼠研究表明西罗莫司剂量増加可降低伤口强度,作者假设这种效应可能是由于接受较高剂量西罗莫司的大鼠体内VEGF和一氧化氮水平较低37。
在一篇综述中,作者得出结论,高剂量mTOR抑制剂不利于伤口愈合,但在低剂量下似乎没有影响38。鉴于mTOR抑制剂和伤口愈合的早期问题,使用此类免疫抑制剂的治疗方案已发展为低剂量mTOR抑制剂和联合治疗。在一项大型TRANSFORM随机对照试验中,研究者在肾移植患者中比较了依维莫司加减量CNI与麦考酚酸加标准剂量CNI(标准治疗)的效果,结果显示伤口相关不良事件无统计学差异(20.6% vs 17.3%;RR 1.19 95% CI 0.99至1.43)39。该研究的一个局限性是需密切监测依维莫司浓度以及难以达到3 ng/mL至8 ng/mL的目标血浆浓度C039。总体而言,对当前文献的回顾提示mTOR抑制剂会对伤口愈合产生不利影响,尤其是在较高剂量下,改善给药方案可能会降低或减轻此类风险。
抗増殖剂
常用的抗増殖剂有三种:吗替麦考酚酯(MMF)和麦考酚钠(MPS)(均为肌苷一磷酸脱氢酶[IMPDH]抑制剂)和硫唑嘌呤2,40。
IMPDH抑制剂的免疫抑制机制与mTOR抑制剂相似。在体内,MMF和MPS转化为麦考酚酸,麦考酚酸可阻断对DNA合成至关重要的部分通路,从而减少T细胞和B细胞増殖2,5,40。MMF具有免疫抑制作用,可用于心脏、肾脏和肺移植3,40。MPS用于肾移植40。硫唑嘌呤用作肾移植和自身免疫性疾病(包括类风湿性关节炎、克罗恩病和多发性硬化)的免疫抑制药物22,41。在体内,硫唑嘌呤与谷胱甘肽反应并转化为6-巯基嘌呤,然后生成额外的代谢物,最终阻断嘌呤合成并刺激T细胞3,22。
一项研究在肾移植受者中比较了两种不同剂量的MMF,结果显示,需要手术干预的伤口发生率无显著差异,同样,接受局部伤口护理的伤口发生率也无显著差异42。基于我们对文章标题的审查(作为检索的一部分),未发现关于硫唑嘌呤对人体外部伤口愈合唯一影响的研究。在一项大鼠研究中比较硫唑嘌呤与安慰剂时,发现硫唑嘌呤组大鼠伤口的愈合时间长于安慰剂组,表明硫唑嘌呤对伤口愈合产生了不利影响,但尚不清楚其对人类的影响程度43。
抗代谢药
甲氨蝶呤是一种常用的叶酸拮抗剂免疫抑制剂,适用于多种风湿病。在较高剂量下也具有抗肿瘤活性。吸收后,其进入细胞并转化为多聚谷氨酸化甲氨蝶呤,竞争性抑制二氢叶酸还原酶,阻止叶酸转化用于构建核酸6。与上述其他药物一样,实验动物研究和体外研究表明药物导致伤口愈合受损,但这些影响并未在临床研究中得到证实,尤其是在术后伤口中6。因此,建议术后继续使用该药物。
皮质类固醇/糖皮质激素
糖皮质激素可阻止细胞因子、细胞粘附分子和补体因子等炎性化学物质的形成3。通过抑制白细胞介素-2的形成,糖皮质激素还可阻止T细胞増殖和活化22。此外,其还会损害单核细胞和B细胞3,22。糖皮质激素是第一种抗排斥药物,但随着时间的推移,其因严重的副作用而逐步淘汰3,22。研究人员一致认为,糖皮质激素对伤口愈合非常不利,会干扰伤口愈合的许多关键阶段,如胶原沉积和合成、血管生成、成纤维细胞増殖、生长因子和吞噬作用等2,3,44,45。
医疗保健提供者的实际考虑因素
免疫系统受损患者(由于药物、合并症或年龄极限)需要额外考虑慢性伤口管理。对于免疫抑制药物,医疗保健提供者不仅应仔细了解用药和给药史(包括剂量变化),还应仔细了解需要使用这些药物的基础疾病史(例如,自身免疫性疾病、器官移植)。由于许多免疫抑制药物均可能损害伤口愈合,医疗保健提供者必须尽早评估伤口愈合潜力,以设定和管理患者期望。考虑到这些患者的复杂性増加,早期转诊至内科或外科专家以协助进行伤口护理以及采取团队合作的方法至关重要。在某些伤口未愈合的情况下,如果无法降低免疫抑制剂的剂量(假设其导致伤口愈合不良),则可能需要设定伤口护理的替代目标(例如,维持或不可愈合),并应咨询其主治医生或专科护理人员。随着有合并症的个体和人群寿命的延长,使用免疫抑制药物治疗有慢性伤口的患者将变得越来越普遍,伤口护理临床医生必须尽早认识到这一点。
讨论
随着医学的不断进步,移植、自身免疫性疾病和恶性肿瘤对免疫抑制的需求也不断増加。在综述中,我们强调了该领域缺乏强有力的研究,并强调了各种免疫抑制对伤口愈合的不同影响。关于糖皮质激素治疗和mTOR治疗(尤其是西罗莫司)对伤口愈合的不利影响,已获得高质量证据。此外,在4项比较西罗莫司与MMF或全身性他克莫司的研究中,所有研究均证实西罗莫司与伤口并发症发生率増加相关35,36,46,47。
关于全身性CNI等药物的文献各不相同,一些认为会对伤口产生不良影响,而另一些则认为会带来获益,因此有必要开展更多直接关注这一问题的研究。新型外用CNI对伤口延迟愈合的影响很小,在某些情况下还会带来获益,但还需对其直接用于慢性伤口进行更多研究。令人欣慰的是,抗増殖药、抗代谢药和新型单克隆抗生素均未显示出愈合减缓的信号。不过,考虑到mAB治疗过程中伤口愈合的证据不足,还需进行更多研究。总体而言,本综述发现,该领域的证据已经过时,围绕免疫抑制剂对伤口愈合的影响得出的结论也不尽相同。此外,我们几乎未发现在人类受试者中的证据。一般而言,在移植后开具免疫抑制剂处方用于预防排斥反应时,患者需服用多种药物才能有效预防排斥反应。因此,在开展单独评价单个药物作用的人体研究方面存在挑战。
由于我们未进行系统综述,某些文章可能被排除在外。在进行检索之前,我们确定了几篇关键综述,并确保这些综述以一种验证形式出现在检索结果中。同时,鉴于该领域的文献较少,尤其是与伤口护理领域相关的文献,叙述性综述在教育和提高服用此类药物的患者的意识方面具有重要价值。
随着免疫抑制需求的不断増加,在该领域开展更多研究至关重要。未来的研究方向包括在动物受试者中使用新型免疫抑制剂进行更多研究,以确定导致伤口延迟愈合的潜在途径和减轻此类影响的潜在方法。此外,还需进行更多高质量研究,对单独和联合免疫治疗进行评价,以更好地了解风险以及不同的免疫抑制剂对伤口愈合的影响。为了探索免疫抑制剂作为慢性或复杂伤口的潜在治疗方法,未来的研究必须在更大的范围内进行,并通过随机对照试验等方法控制临床混杂因素。
结论
综上所述,免疫抑制剂在伤口愈合中可能有益,也可能对其产生显著不利影响。虽然该领域的决定性证据很少,但免疫抑制剂对伤口愈合的影响值得探索,以便更好地为存在慢性伤口不愈合风险或正在经历慢性伤口不愈合的患者量身定制免疫抑制治疗。令人欣慰的是,我们的研究结果表明,并非所有的免疫抑制剂均会产生不利影响,当传统治疗失败时,一些免疫抑制剂可能为伤口治疗带来获益,进而可能提供一种新的伤口治疗选择。
致谢
阿尔伯塔大学高中青年研究员暑期项目。
利益冲突声明
作者声明无利益冲突。
资助
作者未因该项研究收到任何资助。
Author(s)
Aria Appoo
Medical Student, University of Oxford, Oxford, UK
Brandon L Christensen
BMSc MD
Resident Physician, University of Calgary, Division of Infectious Diseases, Calgary, Alberta, Canada
Ranjani Somayaji
BScPT MD MPH
Assistant Professor, University of Calgary, Departments of Medicine, Microbiology, Immunology and Infectious Disease and Community Health Sciences, Calgary, Alberta, Canada
References
- Fireman M, DiMartini AF, Armstrong SC, Cozza KL. Immunosuppressants. Psychosomatics. 2004;45(4). doi:10.1176/appi.psy.45.4.354
- Taylor AL, Watson CJE, Bradley JA. Immunosuppressive agents in solid organ transplantation: Mechanisms of action and therapeutic efficacy. Crit Rev Oncol Hematol. 2005;56(1). doi:10.1016/j.critrevonc.2005.03.012
- Weltz A, Scalea J, Popescu M, Xu J, Bromberg JS. Mechanisms of Immunosuppressive Drugs. In: Kidney Transplantation. Springer New York; 2014. doi:10.1007/978-1-4939-0342-9_12
- Subramanian S, Trence DL. Immunosuppressive Agents: Effects on Glucose and Lipid Metabolism. Endocrinol Metab Clin North Am. 2007;36(4). doi:10.1016/j.ecl.2007.07.003
- Mika A, Stepnowski P. Current methods of the analysis of immunosuppressive agents in clinical materials: A review. J Pharm Biomed Anal. 2016;127. doi:10.1016/j.jpba.2016.01.059
- Pountos I, Giannoudis P V. Effect of methotrexate on bone and wound healing. Expert Opin Drug Saf. 2017;16(5):535-545. doi:10.1080/14740338.2017.1310839
- Willems MCM, van der Vliet JA, Lomme RMLM, Hendriks T. Tacrolimus Does Not Affect Early Wound Healing in a Rodent Model of Bowel Anastomoses and Abdominal Wall Closure. PLoS One. 2013;8(9). doi:10.1371/journal.pone.0076348
- Azzi JR, Sayegh MH, Mallat SG. Calcineurin Inhibitors: 40 Years Later, Can’t Live Without …. The Journal of Immunology. 2013;191(12). doi:10.4049/jimmunol.1390055
- Safarani OA, Patel J. Calcineurin Inhibitors. StatPearls Publishing; 2021.
- Willems MCM, van der Vliet JA, Lomme RMLM, Hendriks T. Tacrolimus Does Not Affect Early Wound Healing in a Rodent Model of Bowel Anastomoses and Abdominal Wall Closure. PLoS One. 2013;8(9). doi:10.1371/journal.pone.0076348
- Schäffer MR, Fuchs N, Proksch B, Bongartz M, Beiter T, Becker HD. TACROLIMUS IMPAIRS WOUND HEALING. Transplantation. 1998;65(6). doi:10.1097/00007890-199803270-00008
- The Effect of Tacrolimus on Lower Extremity Ulcers: A Case Study and Review of the Literature. Wound Manag Prev. 2008;54(4).
- Nemlander A, Ahonen J, Wiktorowicz K, et al. Effect of Cyclosporine on Wound Healing An Analysis With Viscous Cellulose Sponges. Transplantation. 1983;36(1):1-5.
- Petri J, Schurk S, Gebauer S, Haustein U. Cyclosporine A delays wound healing and apoptosis and suppresses activin beta-A expression in rats. European Journal of Dermatology. 1998;8(2):104-113.
- Goldberg M, Lima O, Morgan E, et al. A comparison between cyclosporin A and methylprednisolone plus azathioprine on bronchial healing following canine lung autotransplantation. J Thorac Cardiovasc Surg. 1983;85(6). doi:10.1016/S0022-5223(19)37469-0
- Sussman G. The use of topical calcineurin inhibitors in chronic wound management. Wound Practice & Research: Journal of the Australian Wound Management Association. 2000;26(3):140-145.
- Frazier W, Bhardwaj N. Atopic Dermatitis: Diagnosis and Treatment. Am Fam Physician. 2020;101(10):590-598.
- Lyon C, Stapleton M, Smith A, Mendelsohn S, Beck M, Griffiths C. Topical tacrolimus in the management of peristomal pyoderma gangrenosum. Journal of Dermatological Treatment. 2001;12(1). doi:10.1080/095466301750163518
- Ginocchio L, Draghi L, Darvishian F, Ross FL. Refractory Ulcerated Necrobiosis Lipoidica: Closure of a Difficult Wound with Topical Tacrolimus. Adv Skin Wound Care. 2017;30(10). doi:10.1097/01.ASW.0000521867.98577.a5
- Mackelfresh J, Soon S, Arbiser JL. Combination Therapy of Doxycycline and Topical Tacrolimus for Venous Ulcers. JAMA Dermatol. 2005;141(11). doi:10.1001/archderm.141.11.1476
- Namkoong S, Chung J, Yoo J, et al. Topical tacrolimus does not negatively impact acute skin wound healing. Exp Dermatol. 2013;22(5). doi:10.1111/exd.12131
- Khan MM. Immunopharmacology. Springer US; 2008. doi:10.1007/978-0-387-77976-8
- Bibbo C, Goldberg JW. Infectious and Healing Complications after Elective Orthopaedic Foot and Ankle Surgery during Tumor Necrosis Factor-Alpha Inhibition Therapy. Foot Ankle Int. 2004;25(5). doi:10.1177/107110070402500510
- Streit M, Beleznay Z, Braathen LR. Topical application of the tumour necrosis factor-alpha antibody infliximab improves healing of chronic wounds. Int Wound J. 2006;3(3). doi:10.1111/j.1742-481X.2006.00233.x
- Hewitt D, Tait C. Use of infliximab in pyoderma gangrenosum. Australasian Journal of Dermatology. 2007;48(2). doi:10.1111/j.1440-0960.2007.00344.x
- Fox JD, Baquerizo-Nole KL, Keegan BR, et al. Adalimumab treatment leads to reduction of tissue tumor necrosis factor-alpha correlated with venous leg ulcer improvement: a pilot study. Int Wound J. 2016;13(5). doi:10.1111/iwj.12497
- Lopes JV, Freitas LAM de, Marques RD, Bocca AL, Sousa JB de, Oliveira PG de. Analysis of the tensile strength on the healing of the abdominal wall of rats treated with infliximab. Acta Cir Bras. 2008;23(5). doi:10.1590/S0102-86502008000500009
- Larrañaga MD, Lewis S, Richard J, Robert A, eds. Polyclonal Antibodies. In: Hawley’s Condensed Chemical Dictionary. 16th ed. Wiley; 2016.
- Johnson M. Monoclonal Antibodies. In: Longe JL, ed. Gale Encyclopedia of Nursing and Allied Health. 4th ed. Credo Reference; 2018.
- Ueno P, Felipe C, Ferreira A, et al. Wound Healing Complications in Kidney Transplant Recipients Receiving Everolimus. Transplantation. 2017;101(4). doi:10.1097/TP.0000000000001392
- Patel S, Pankewycz O, Kohli R, et al. Obesity in Renal Transplantation: The Role of Induction Therapy on Long-Term Outcomes. Transplant Proc. 2011;43(2). doi:10.1016/j.transproceed.2011.01.040
- Kordestani SS. Atlas of Wound Healing. (Abyaneh S, Fayyazbakhsh, eds.). Elsevier; 2019.
- Azzola A, Havryk A, Chhajed P, et al. Everolimus and mycophenolate mofetil are potent inhibitors of fibroblast proliferation after lung transplantation1. Transplantation. 2004;77(2). doi:10.1097/01.TP.0000101822.50960.AB
- P. B. Wound healing and the role of fibroblasts. J Wound Care. 2013;22(8). doi:10.12968/jowc.2013.22.8.407
- Dean PG, Lund WJ, Larson TS, et al. Wound-Healing Complications After Kidney Transplantation: A Prospective, Randomized Comparison Of Sirolimus And Tacrolimus1. Transplantation. 2004;77(10). doi:10.1097/01.TP.0000123082.31092.53
- Larson TS, Dean PG, Stegall MD, et al. Complete Avoidance of Calcineurin Inhibitors in Renal Transplantation: A Randomized Trial Comparing Sirolimus and Tacrolimus. American Journal of Transplantation. 2006;6(3). doi:10.1111/j.1600-6143.2005.01177.x
- Schäffer M, Schier R, Napirei M, Michalski S, Traska T, Viebahn R. Sirolimus impairs wound healing. Langenbecks Arch Surg. 2007;392(3). doi:10.1007/s00423-007-0174-5
- Nashan B, Citterio F. Wound Healing Complications and the Use of Mammalian Target of Rapamycin Inhibitors in Kidney Transplantation. Transplantation. 2012;94(6). doi:10.1097/TP.0b013e3182551021
- Citterio F, Henry M, Kim DY, et al. Wound healing adverse events in kidney transplant recipients receiving everolimus with reduced calcineurin inhibitor exposure or current standard-of-care: Insights from the 24 month TRANSFORM study. Expert Opin Drug Saf. Published online 2020:1339-1348. doi:10.1080/14740338.2020.1792441
- Staatz CE, Tett SE. Clinical Pharmacokinetics and Pharmacodynamics of Mycophenolate in Solid Organ Transplant Recipients. Clin Pharmacokinet. 2007;46(1). doi:10.2165/00003088-200746010-00002
- Casetta I, Iuliano G, Filippini G. Azathioprine for multiple sclerosis. Cochrane Database of Systematic Reviews. Published online October 17, 2007. doi:10.1002/14651858.CD003982.pub2
- Flechner SM, Feng J, Mastroianni B, et al. The Effect of 2-Gram Versus 1-Gram Concentration Controlled Mycophenolate Mofetil on Renal Transplant Outcomes Using Sirolimus-Based Calcineurin Inhibitor Drug-Free Immunosuppression. Transplantation. 2005;79(8). doi:10.1097/01.TP.0000155430.52845.51
- Ginestal R, Pérez-Köhler B, Pérez-López P, et al. Comparing the influence of two immunosuppressants (fingolimod, azathioprine) on wound healing in a rat model of primary and secondary intention wound closure. Wound Repair and Regeneration. 2019;27(1). doi:10.1111/wrr.12685
- Wicke C. Effects of Steroids and Retinoids on Wound Healing. Archives of Surgery. 2000;135(11). doi:10.1001/archsurg.135.11.1265
- Anstead G. Steroids, retinoids, and wound healing. Adv Wound Care (New Rochelle). 1998;11(6):277-285.
- Valente JF, Hricik D, Weigel K, et al. Comparison of Sirolimus vs. Mycophenolate Mofetil on Surgical Complications and Wound Healing in Adult Kidney Transplantation. American Journal of Transplantation. 2003;3(9). doi:10.1034/j.1600-6143.2003.00185.x
- Kuppahally S, Al-Khaldi A, Weisshaar D, et al. Wound Healing Complications with De Novo Sirolimus Versus Mycophenolate Mofetil-Based Regimen in Cardiac Transplant Recipients. American Journal of Transplantation. 2006;6(5p1). doi:10.1111/j.1600-6143.2006.01282.x
- Ormerod AD, Thomas KS, Craig FE, et al. Comparison of the two most commonly used treatments for pyoderma gangrenosum: results of the STOP GAP randomised controlled trial. BMJ. 2015;350(jun12 3). doi:10.1136/bmj.h2958
- Flechner SM, Zhou L, Derweesh I, et al. The impact of sirolimus, mycophenolate mofetil, cyclosporine, azathioprine, and steroids on wound healing in 513 kidney-transplant recipients. Transplantation. 2003;76(12). doi:10.1097/01.TP.0000093502.26208.42
- Citterio F, Henry M, Kim DY, et al. Wound healing adverse events in kidney transplant recipients receiving everolimus with reduced calcineurin inhibitor exposure or current standard-of-care: insights from the 24-month TRANSFORM study. Expert Opin Drug Saf. 2020;19(10). doi:10.1080/14740338.2020.1792441
- Kapugi M, Cunningham K. Corticosteroids. Orthopaedic Nursing. 2019;38(5):336-339. doi:10.1097/NOR.0000000000000595
- Ponticelli C. Cyclosporine: From Renal Transplantation to Autoimmune Diseases. Ann N Y Acad Sci. 2005;1051(1):551-558. doi:10.1196/annals.1361.099
- Akar Y, Yucel G, Durukan AH, Yucel I, Arici G. Laboratory Science. Systemic toxicity of tacrolimus given by various routes and the response to dose reduction. Clin Exp Ophthalmol. 2005;33(1):53-59. doi:10.1111/j.1442-9071.2005.00942.x
- Kurian K, Addisu A. Sirolimus: A Novel Immunosuppressive Drug in Heart Transplantation. Recent Pat Cardiovasc Drug Discov. 2009;4(3):187-191. doi:10.2174/157489009789152294
- Sehgal SN. Sirolimus: its discovery, biological properties, and mechanism of action. Transplant Proc. 2003;35(3):S7-S14. doi:10.1016/S0041-1345(03)00211-2
- van Gelder T, Fischer L, Shihab F, Shipkova M. Optimizing everolimus exposure when combined with calcineurin inhibitors in solid organ transplantation. Transplant Rev. 2017;31(3):151-157. doi:10.1016/j.trre.2017.02.007
- Falkowski S, Woillard JB. Therapeutic Drug Monitoring of Everolimus in Oncology: Evidences and Perspectives. Ther Drug Monit. 2019;41(5):568-574. doi:10.1097/FTD.0000000000000628
- Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury.; 2020.
- Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury.; 2017. Accessed February 10, 2022. https://www-ncbi-nlm-nih-gov.ezproxy.lib.ucalgary.ca/books/NBK548018/
- Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury.; 2017. Accessed February 10, 2022. https://www-ncbi-nlm-nih-gov.ezproxy.lib.ucalgary.ca/books/NBK548587/
- Lichtenstein L, Ron Y, Kivity S, et al. Infliximab-Related Infusion Reactions: Systematic Review. J Crohns Colitis. 2015;9(9):806-815. doi:10.1093/ecco-jcc/jjv096
- Tracey D, Klareskog L, Sasso EH, Salfeld JG, Tak PP. Tumor necrosis factor antagonist mechanisms of action: A comprehensive review. Pharmacol Ther. 2008;117(2):244-279. doi:10.1016/j.pharmthera.2007.10.001
- Chavez-Alvarez S, Herz-Ruelas M, Villarreal-Martinez A, Ocampo-Candiani J, Garza-Garza R, Gomez-Flores M. Azathioprine: its uses in dermatology. An Bras Dermatol. 2020;95(6):731-736. doi:10.1016/j.abd.2020.05.003


