Volume 18 Number 1
Implementation of exercise therapy in kidney failure
Rachel Zui Chih Teo, Derlinder Kaur, Douglas Ong, Tripti Singh and Behram Ali Khan
Keywords dialysis, quality of life, exercise therapy, functional ability
For referencing Teo RZC. Implementation of exercise therapy in kidney failure. Renal Society of Australasia Journal 2022; 18(1):9-14
DOI
https://doi.org/10.33235/rsaj.18.1.9-14
Submitted 25 October 2021
Abstract
Background Low levels of physical activity in patients with chronic kidney disease have long been shown to impact adversely on functional status and quality of life. Numerous studies have demonstrated the benefits of exercise in patients on haemodialysis. However, implementation of exercise programs as routine practice remains lacking, likely due to the variability of methodology across published studies as well as limitations at dialysis facilities.
Objectives We share our experience in providing exercise therapy that is prioritised as routine patient care in a community dialysis setting; interventions are individualised to each patient’s functional ability based on thorough and regular assessment.
Approach Exercise therapy is structured whereby patients’ functional status and abilities are evaluated in detail prior to placement into exercise programs and at regular progress intervals. Each exercise program, facilitated by exercise specialists, is tailored to the patients’ functional capability and health status using exercises that are modifiable to their needs. Group exercises also provide an avenue of social interaction and support for patients.
Conclusion Exercise therapy is advocated strongly to improve function and quality of life in patients on dialysis. Through implementation of a range of multifaceted and individualised exercise programs, we address both their functional and psychosocial needs. Our unique holistic approach aims to facilitate patient adherence in a sustainable manner.
Introduction
Patients with chronic kidney disease (CKD) commonly exhibit low levels of physical activity and poor physical functions due to uraemic myopathy, neuropathy and anaemia, in addition to sedentary lifestyle and ageing (Zelle et al., 2017). This in turn results in reduced muscle strength, increased cardiovascular risk, and reduced physical fitness (Anding et al., 2015; Zelle et al., 2017).
Both physical and mental components contribute to the poor health-related quality of life (QOL) in patients with CKD. Feelings of anxiety and depression result in heightened feelings of helplessness which can amplify negative aspects of their condition and lead to a deterioration of physical function (Cukor et al., 2007; Goh & Griva, 2018). These feelings may also arise when patients are unwilling to recognise or accept their conditions (Verhoof et al., 2014). Hence, it is paramount to provide both social and physical support for patients.
Research has shown that exercise is a safe and effective rehabilitative intervention for patients with end-stage kidney disease (ESKD) and improves physical function, QOL and dialysis efficacy (Clarkson et al., 2019; Huang et al., 2019; Salhab et al., 2019). Additionally, there are mental health benefits such as reduced depression and stress, and an increased sense of accomplishment (Jhamb et al., 2016). Indeed, the National Kidney Foundation Kidney Disease Outcomes Quality Initiative Guidelines formally recommend that all dialysis patients should be counselled regularly by nephrology and dialysis staff to increase their level of physical activity, and that physical functioning assessment and encouragement to participate in physical activity should be part of routine patient care (K/DOQI Workgroup, 2005).
However, prior studies have shown that assessment of physical function is not part of routine practice (Painter et al., 2014), nor are dialysis staff or nephrologists confident in counselling patients about exercise (Delgado & Johansen, 2010). Barriers to exercise are truly global and are related to policy and practice (Bennett et al., 2021). Implementation of exercise programs remains variable and limited, possibly due to differences in exercise modalities (in-centre inter- or intra-dialytic versus home-based), intensity and duration found across published studies. This highlights the need for change in practice patterns, and emphasises the importance of the role of exercise specialists who are specially trained in exercise prescription.
The National Kidney Foundation (NKF) Singapore, the largest voluntary welfare organisation providing dialysis care in the community for over 4000 patients in Singapore, advocates active, healthy living to maintain functional ability and improve QOL in patients on dialysis. Exercise specialists from the Exercise and Fitness (ENF) Department conceptualise and facilitate various exercise programs focused on reversing sedentary lifestyles that are prevalent in dialysis patients, and also provide community outreach interventions. Exercise specialists employed by NKF Singapore are individuals who have graduated with a major in sports science, a multidisciplinary field of study covering subjects such as anatomy and physiology, sports psychology and biomechanics which look at the human body’s reaction to stimuli such as sports and exercise.
Here, we share the current exercise therapy program implemented for our patients, consisting mostly of patients on haemodialysis and a small number on peritoneal dialysis. Our program structure differs largely from that of community dialysis facilities in other countries; the uniqueness lies in the tailored approach under a holistic care model which caters to each patient’s assessed functional ability in order to facilitate adherence and sustain long-term goals. It has been shown that an engaging and individualised exercise program catering to the patient’s health status and ability facilitates long-term adherence (Jhamb et al., 2016).
Exercise program pre-placement assessment
The Dialysis Placement Clinic (DPC) is a multidisciplinary platform that allows opportune evaluation of patients’ status prior to placement into dialysis centres. Exercise specialists evaluate patients’ functional capacity and independence using handgrip assessment, modified Short Physical Performance Battery (SPPB) (Guralnik et al., 1994) and Performance Oriented Mobility Assessment (POMA) (Tinetti et al., 1986).
Handgrip strength is widely used as an indicator of frailty in the elderly given its simple, fast and non-invasive application while being a reliable marker of malnutrition (Vogt et al., 2016). In patients with CKD, lower handgrip strength correlates with higher mortality rate. There is no consensus on the cut-off values to predict mortality in dialysis patients – values of 28.3kg and 21.5kg for male and females respectively have been suggested as the best indicators (Matos et al., 2014), while lower and more disparate cut-off values of 22.5kg in males and 7kg in females were found in a different study (Vogt et al., 2016). The study by Matos et al. consisted of mostly non‑White patients (88.9%), while this was unreported in the study by Vogt et al. which also consisted older patients (mean of 58 years old versus 46 years old). Research has shown that patients on dialysis are able to achieve handgrip strength values ranging from 12–38kg in males and 12–26kg in females (Leal et al., 2011). We utilise the grip strength component of the Physical Frailty Phenotype (PFP), given its values fall within the reported capabilities of dialysis patients (Fried et al., 2001) and it is the most commonly applied, well validated frailty metric in the CKD and dialysis population (Chowdhury et al., 2017). Grip strength is scaled to the body mass index (BMI) of the patient (Table 1).
Table 1. Criteria for grip strength assessment, extracted from the PFP (Fried et al., 2001)
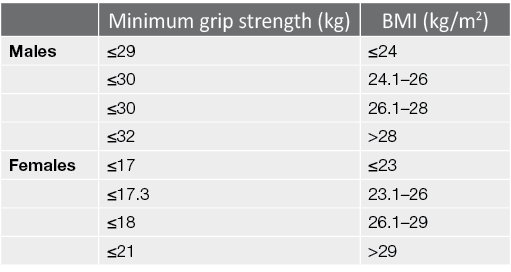
The modified SPPB assessment and Tinetti POMA are used to evaluate a patient’s capacity in performing activities of daily living (ADLs) and ambulating in the community. The SPPB assessment comprises of three components – a 3-stage balance test, a 5-time sit-to-stand (STS) and a gait speed test. Due to space constraints, we replaced the gait speed test with the Tinetti POMA.
The 3-stage balance test involves the patient balancing for 10 seconds in each of three positions that include feet-together, half-tandem and full-tandem. The Center for Disease Control and Prevention (CDC) suggests that inability to hold oneself in a tandem balance for 10 seconds is indicative of a higher fall risk (CDC, 2021).
In the 5-time STS assessment, patients perform 5 repetitions of STS as quickly as possible (Guralnik et al., 1994). It has been reported that for every 1mL/min per 1.73m2 decrease in glomerular filtration rate, the inability to complete one unassisted rise was 1.5 times greater, with the odds increasing for patients with diabetes (Painter & Marcus, 2013). Additionally, the risk of recurrent falls tend to be higher, with a 74% increase in community-dwelling elderly when they took 15 seconds or more to complete the STS assessment (Buatois et al., 2008).
POMA is a commonly used screening tool appropriate for identifying the risk of falls in older adults in both research and clinical settings (Jahantabi-Nejad & Azad, 2019), comprising 16 items of different manoeuvres, which test an individual’s balance, postural control and gait.
Results from the DPC assessment, together with the inclusion/exclusion criteria (Table 2), are applied to allocate patients to the appropriate exercise program.
Table 2. Medical exclusion and inclusion criteria for various exercise programs

Exercise programs facilitated by ENF
The current exercise therapy programs implemented for our patients are outlined below and summarised in Figure 1. These are divided into three pathways – in-centre, via an integrated team care model, and through our outreach program.
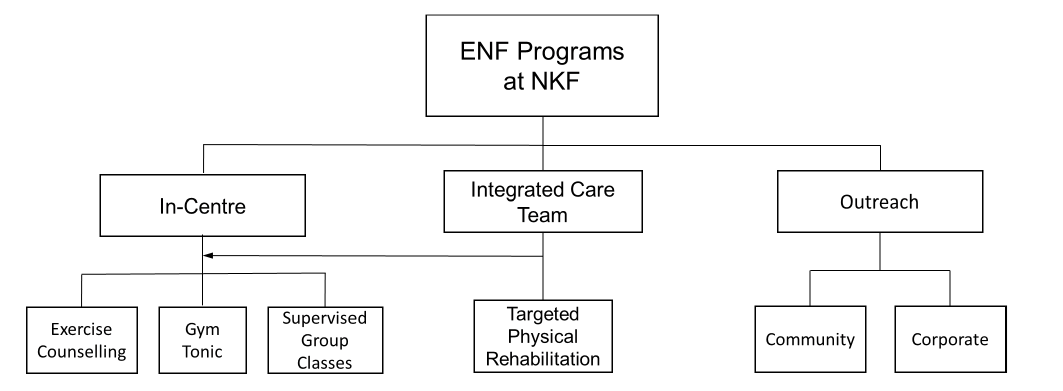
Figure 1. Structure of exercise programs provided by the ENF department at NKF, which includes in-centre programs for patients as well as outreach programs which cater to the wider community
In-centre
Exercise counselling
This program was established in 2014 and is conducted via physical consultations at dialysis centres or teleconsultations. All patients are reviewed by the ENF team upon admission into NKF. Frequency of review depends on patients’ overall risk, pre-set goals and urgency of intervention. The review interval ranges from 1 month for high risk patients, to 6–9 months for lower risk patients. Exercise specialists educate patients on the importance of leading an active lifestyle and on various methods to increase their daily activity level. Simultaneously, their mobility status and independence level for ADLs are reviewed. Exercise specialists then tailor specific programs catering to the patient’s functional capability and medical status. The aim, particularly for the elderly, is to delay the progression of muscle atrophy and reduce the risk of falls (Santanasto et al., 2017; Thomas et al., 2019).
Tailored exercise programs form a unique aspect of this service. The patient’s perspective is considered, allowing patient autonomy, as a one-size-fits-all approach to exercise prescription is likely inadequate in addressing the myriad of health concerns in patients on dialysis.
Gym Tonic
Project Gym Tonic, initiated in collaboration with the Lien Foundation and the University of Finland in 2018, is carried out at NKF’s integrated renal centre. The 12-week strength training program uses the principle of progressive overload to maintain and/or increase muscle strength to reduce the incidence of falls and degree of frailty. Higher muscle mass and grip strength has been associated with lower mortality rates in patients with ESKD (Watson et al., 2020).
The Gym Tonic program uses six of HUR’s (Finland) pneumatic machines – comprising of leg press, abdomen/back, leg extension/curl, leg abduction/adduction, chest press and lateral pull – which target the body’s major muscle groups. The program is individualised and prescribed for each patient using a baseline WELMED assessment (Björkgren et al., 2021), and a repeat assessment at week 13 to ascertain the patient’s progress. The WELMED assessment includes anthropometry, grip strength, walking, STS and isometric muscle tests, as well as lifestyle and pain assessment questionnaires. Progress assessments allow re-evaluation of the patient’s rehabilitation needs to improve individualised care planning and intervention.
Progressive strength training programs should be implemented conservatively in patients with ESKD. As such, the Gym Tonic program has specific inclusion criteria (Table 2) for identifying suitable patients.
Supervised group classes
Initiated in 2014, this program aims to improve overall physical function. Group classes are supervised at a 1:4 exercise specialist to patient ratio. Patients are required to remain seated for majority of the class to reduce the risk of falls. This allows more ESKD patients, including the more frail, to participate. A warm-up is done by seated aerobics, and cool-down by seated yoga and static stretching. Exercises are modified into versions that can be performed seated on a stable chair. Patients are able to take home various parts of exercises which they feel comfortable performing at home. Group classes also promote social cohesion among patients. During the COVID‑19 pandemic, virtual platforms were established, but similar exercise modalities remained. This allowed patients to stay active and maintain social interaction to reduce the risk of social isolation during the pandemic.
Both the Gym Tonic and supervised group classes serve as platforms for social interaction and support while sharing knowledge with patients. Regular interaction with patients also allowed the ENF team to make timely referrals to other allied health professionals as required.
Integrated care team model
The integrated care team (ICT) model consists of doctors, nurses, social workers, dietitians, occupational therapists (OT) and exercise specialists. The team works together to provide holistic care for patients on dialysis; complex cases are provided prompt care. Frequency of case reviews are based on the complexity of issues.
The ENF department focuses on patients’ physical function. Patients with higher physical function and independence are steered to Gym Tonic or supervised group classes, while those with lower physical function are allocated to targeted physical rehabilitation. Targeted physical rehabilitation focuses on specific weaknesses. Exercise specialists work with the OT and the patient towards achieving specific goals. For example, increasing lower limb strength to increase standing/walking tolerance and eventually walking distance. This program utilises minimal equipment and more of body weight. Exercises and intensity are modified to each patient’s needs.
Outreach program
NKF outreach programs provide public education to reinforce preventive measures that can reduce the risk of developing CKD. We reach out to youths in schools and working adults in the corporate sector, delivering face-to-face or virtual talks focused largely on lifestyle factors attributed to the cause of CKD. The ENF team promotes the beneficial effects of exercise as both prevention and disease management. Key exercises and sample training protocols are also shared during sessions.
Through education, we aim to empower individuals with the knowledge and ability to make lifestyle changes such as healthier food choices, and educate them with a habit of regular exercise in order to reduce the risk of CKD.
Outcomes
Currently we have 1970 patients who are actively participating in one or more of the exercise programs run by exercise specialists from ENF at NKF Singapore. This makes up 49.3% of our prevalent patients on haemodialysis and peritoneal dialysis. In this descriptive report, we share our experience in developing individualised exercise therapy programs over the years, and demonstrate the plausibility of implementation of such programs in patients on dialysis in the community. Although we are yet to have objective data to substantiate the effectiveness of our programs, collation of functional data is underway. We aim to share our findings subsequently to demonstrate the value of individualised exercise therapy in patients on dialysis.
Conclusion
Exercise therapy is advocated strongly to improve function and QOL in patients on dialysis. Here we share the multifaceted, individualised exercise programs provided for patients using a holistic approach to address both their functional and psychosocial needs in a sustainable manner. Exercise should also be promoted in the wider community as preventive medicine.
Conflict of interest
The authors declare no conflicts of interest.
Funding
The authors received no funding for this study.
Author(s)
Dr Rachel Zui Chih Teo1 MBBS (Hons), FRACP, Consultant
Ms Derlinder Kaur2 BSci (Sports and Exercise Science), Exercise Specialist
Mr Douglas Ong2 BSci (Sports and Exercise Science), Exercise Specialist
Dr Tripti Singh1 MBBS, MRCP (UK), Consultant
Dr Behram Ali Khan1 MBBS, MD (USA), Medical Director
1 Department of Medical Services, The National Kidney Foundation, Singapore
2 Department of Exercise and Fitness, The National Kidney Foundation, Singapore
Correspondence to Rachel Teo, The National Kidney Foundation, 81 Kim Keat Road, Singapore 328836
Email rachel.teo@nkfs.org
References
Anding, K., Bär, T., Trojniak-Hennig, J., Kuchinke, S., Krause, R., Rost, J. M., & Halle, M. (2015). A structured exercise programme during haemodialysis for patients with chronic kidney disease: Clinical benefit and long-term adherence. BMJ Open, 5(8), e008709. doi:10.1136/bmjopen-2015-008709
Bennett, P. N., Kohzuki, M., Bohm, C., Roshanravan, B., Bakker, S. J. L., Viana, J. L., MacRae, J. M., Wilkinson, T. J., Wilund, K. R., Van Craenenbroeck, A. H., Sakkas, G. K., Mustata, S., Fowler, K., McDonald, J., Aleamañy, G. M., Anding, K., Avin, K. G., Escobar, G. L., Gabrys, I., . . . Thompson, S. (2021). Global policy barriers and enablers to exercise and physical activity in kidney care. Journal of Renal Nutrition. doi:10.1053/j.jrn.2021.06.007
Björkgren, M., Borg, F., Tan, K., Laxåback, G., & Nygård, L. (2021). Introducing progressive strength training program in Singapore’s elder care settings [original research]. Frontiers in Medicine, 8(1163). doi:10.3389/fmed.2021.515898
Buatois, S., Miljkovic, D., Manckoundia, P., Gueguen, R., Miget, P., Vançon, G., Perrin, P., & Benetos, A. (2008). Five times sit to stand test is a predictor of recurrent falls in healthy community-living subjects aged 65 and older. Journal of the American Geriatric Society, 56(8), 1575–1577. doi:10.1111/j.1532-5415.2008.01777.x
CDC. (2021, July 26). STEADI: Older adult fall prevention. Retrieved from https://www.cdc.gov/steadi/pdf/4-Stage_Balance_Test
Chowdhury, R., Peel, N. M., Krosch, M., & Hubbard, R. E. (2017). Frailty and chronic kidney disease: A systematic review. Archives of Gerontology & Geriatrics, 68, 135–142. doi:10.1016/j.archger.2016.10.007
Clarkson, M. J., Bennett, P. N., Fraser, S. F., & Warmington, S. A. (2019). Exercise interventions for improving objective physical function in patients with end-stage kidney disease on dialysis: A systematic review and meta-analysis. American Journal of Physiology Renal Physiology, 316(5), F856-F872. doi:10.1152/ajprenal.00317.2018
Cukor, D., Cohen, S. D., Peterson, R. A., & Kimmel, P. L. (2007). Psychosocial aspects of chronic disease: ESRD as a paradigmatic illness. Journal of the American Society of Nephrology, 18(12), 3042–3055. doi:10.1681/asn.2007030345
Delgado, C., & Johansen, K. L. (2010). Deficient counseling on physical activity among nephrologists. Nephron Clinical Practice, 116(4), c330–336. doi:10.1159/000319593
Fried, L. P., Tangen, C. M., Walston, J., Newman, A. B., Hirsch, C., Gottdiener, J., Seeman, T., Tracy, R., Kop, W. J., Burke, G., McBurnie, M. A., & Cardiovascular Health Study Collaborative Research Group. (2001). Frailty in older adults: Evidence for a phenotype. The Journals Of Gerontology. Series A, Biological Sciences and Medical Sciences, 56(3), M146–156. doi:10.1093/gerona/56.3.m146
Goh, Z. S., & Griva, K. (2018). Anxiety and depression in patients with end-stage renal disease: Impact and management challenges – a narrative review. International Journal of Nephrology & Renovascular Disease, 11, 93–102. doi:10.2147/IJNRD.S126615
Guralnik, J. M., Simonsick, E. M., Ferrucci, L., Glynn, R. J., Berkman, L. F., Blazer, D. G., Scherr, P. A., & Wallace, R. B. (1994). A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. Journal of Gerontology 49(2), M85–94. doi:10.1093/geronj/49.2.m85
Huang, M., Lv, A., Wang, J., Xu, N., Ma, G., Zhai, Z., Zhang, B., Gao, J., & Ni, C. (2019). Exercise training and outcomes in hemodialysis patients: Systematic review and meta-analysis. American Journal of Nephrology, 50(4), 240–254. doi:10.1159/000502447
Jahantabi-Nejad, S., & Azad, A. (2019). Predictive accuracy of performance oriented mobility assessment for falls in older adults: A systematic review. Medical Journal of the Islamic Republic of Iran, 33, 38. doi:10.34171/mjiri.33.38
Jhamb, M., McNulty, M. L., Ingalsbe, G., Childers, J. W., Schell, J., Conroy, M. B., Forman, D. E., Hergenroeder, A., & Dew, M. A. (2016). Knowledge, barriers and facilitators of exercise in dialysis patients: A qualitative study of patients, staff and nephrologists. BMC Nephrology, 17(1), 192. doi:10.1186/s12882-016-0399-z
K/DOQI Workgroup. (2005). K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. American Journal of Kidney Disease, 45(4 Suppl 3), S1–153. https://www.ncbi.nlm.nih.gov/pubmed/15806502
Leal, V. O., Mafra, D., Fouque, D., & Anjos, L. A. (2011). Use of handgrip strength in the assessment of the muscle function of chronic kidney disease patients on dialysis: A systematic review. Nephrology, Dialysis, Transplantation, 26(4), 1354–1360. doi:10.1093/ndt/gfq487
Matos, C. M., Silva, L. F., Santana, L. D., Santos, L. S., Protásio, B. M., Rocha, M. T., Ferreira, V. L., Azevedo, M. F., Martins, M. T., Lopes, G. B., & Lopes, A. A. (2014). Handgrip strength at baseline and mortality risk in a cohort of women and men on hemodialysis: A 4-year study. Journal of Renal Nutrition, 24(3), 157–162. doi:10.1053/j.jrn.2013.12.005
Painter, P., Clark, L., & Olausson, J. (2014). Physical function and physical activity assessment and promotion in the hemodialysis clinic: A qualitative study. American Journal of Kidney Disease, 64(3), 425–433. doi:10.1053/j.ajkd.2014.01.433
Painter, P., & Marcus, R. L. (2013). Assessing physical function and physical activity in patients with CKD. Clinical Journal of the American Society of Nephrology, 8(5), 861–872. doi:10.2215/cjn.06590712
Salhab, N., Karavetian, M., Kooman, J., Fiaccadori, E., & El Khoury, C. F. (2019). Effects of intradialytic aerobic exercise on hemodialysis patients: A systematic review and meta-analysis. Journal of Nephrology, 32(4), 549–566. doi:10.1007/s40620-018-00565-z
Santanasto, A. J., Glynn, N. W., Lovato, L. C., Blair, S. N., Fielding, R. A., Gill, T. M., Guralnik, J. M., Hsu, F. C., King, A. C., Strotmeyer, E. S., Manini, T. M., Marsh, A. P., McDermott, M. M., Goodpaster, B. H., Pahor, M., Newman, A. B., & LIFE Study Group. (2017). Effect of physical activity versus health education on physical function, grip strength and mobility. Journal of the American Geriatric Society, 65(7), 1427–1433. doi:10.1111/jgs.14804
Thomas, E., Battaglia, G., Patti, A., Brusa, J., Leonardi, V., Palma, A., & Bellafiore, M. (2019). Physical activity programs for balance and fall prevention in elderly: A systematic review. Medicine (Baltimore), 98(27), e16218. doi:10.1097/md.0000000000016218
Tinetti, M. E., Williams, T. F., & Mayewski, R. (1986). Fall risk index for elderly patients based on number of chronic disabilities. American Journal of Medicine, 80(3), 429–434. doi:10.1016/0002-9343(86)90717-5
Verhoof, E. J., Maurice-Stam, H., Heymans, H. S., Evers, A. W., & Grootenhuis, M. A. (2014). Psychosocial well-being in young adults with chronic illness since childhood: The role of illness cognitions. Child and Adolescent Psychiatry & Mental Health, 8, 12. doi:10.1186/1753-2000-8-12
Vogt, B. P., Borges, M. C. C., Goés, C. R., & Caramori, J. C. T. (2016). Handgrip strength is an independent predictor of all-cause mortality in maintenance dialysis patients. Clinical Nutrition, 35(6), 1429–1433. doi:10.1016/j.clnu.2016.03.020
Watson, E. L., Major, R. W., Wilkinson, T. J., Greening, N. J., Gould, D. W., Barratt, J., & Smith, A. C. (2020). The association of muscle size, strength and exercise capacity with all-cause mortality in non-dialysis-dependent CKD patients. Clinical Physiology & Functional Imaging, 40(6), 399–406. doi:10.1111/cpf.12655
Zelle, D. M., Klaassen, G., van Adrichem, E., Bakker, S. J., Corpeleijn, E., & Navis, G. (2017). Physical inactivity: A risk factor and target for intervention in renal care. Nature Reviews Nephrology, 13(3), 152–168. doi:10.1038/nrneph.2016.187
