Volume 5 Issue 2
Poster abstracts from the avas 2019 National Scientific Meeting
The following poster abstracts were presented at AVAS’ 2019 National Scientific meeting in May and are published here to keep you up-to-date with the latest research results in the vascular access field. If you see something of interest and want to know more, get in touch with the corresponding author (underlined and in bold).
SURVEY OF INDURATION DEVELOPMENT FOLLOWING ANTI-CANCER DRUG ADMINISTRATION THROUGH A PERIPHERAL INTRAVENOUS CATHETER AND RISK FACTORS BY ULTRASONOGRAPHY
Mrs Mari Abe, PhD1; Ryoko Murayama, PhD1,2; Koichi Yabunaka, RN2,3; Mie Tsuge, MNS4; Chieko Komiyama PhD4; Hiromi Sanada2,5
1Department of Advanced Nursing Technology, Graduate School of Medicine, The University of Tokyo, Bunkyo-ku, Japan
2Global Nursing Research Center, Graduate School of Medicine, The University of Tokyo, Bunkyo-ku, Japan
3Department of Imaging Nursing Science, Graduate School of Medicine, The University of Tokyo, Bunkyo-ku, Japan
4Department of Nursing, The University of Tokyo Hospital, Bunkyo-ku, Japan
5Department of Gerontological Nursing/ Wound Care Management, Graduate School of Medicine, The University of Tokyo, Bunkyo-ku, Japan
Introduction Following anticancer drug infusion, the degeneration of subcutaneous tissue, induration, may occur. If induration develops, the site is avoided for catheterization and less suitable sites, including joints or the hands, must be used owing to fewer sites available. The development of induration should be prevented; however, the incidence and risk factors remain unclear.
Objectives The present study was conducted to survey induration incidence and risk factors.
Methods The setting of this cross-sectional observation survey was a chemotherapy room. The sites of anticancer drug infusion administration were recorded, and these were observed the day following administration. Clinical nurses judged the presence or absence of induration by palpation. The sites were observed using ultrasonography (B-mode and elastography). Vein diameter was measured from a B-mode image with image processing software. Logistic regression analysis was performed to analyse induration risk factors.
Results In total, 70 patients were analysed. The induration incidence was 18.6% (13/70), and three abnormal cases were confirmed through ultrasonography findings, including edema, thrombosis, and vessel wall thickness. Age, vein diameter, and fosaprepitant use were included in the logistic regression model with induration as the outcome to calculate odds ratios (ORs) and 95% confidence intervals (CIs). Age: OR 0.93:95, CI 0.87–0.99; vein diameter: OR 0.38:95, CI 0.16–0.91; fosaprepitant use: OR 1.44:95, CI 0.21–9.94. Induration prevalence was significantly lower in older patients or those with larger vein diameters.
Conclusion The results suggest that induration may be prevented by selecting larger diameter vessels using ultrasonography, when catheterizing for anticancer drug administration.
EXTENDED DWELL PERIPHERAL CATHETERS IN PATIENTS WITH DIFFICULT VENOUS ACCESS: DEVELOPMENT OF AN ULTRASOUND GUIDED INSERTION ALGORITHM FOR EXTENDING DWELL OF PERIPHERAL CATHETERS AND REDUCING FAILURE
Dr Evan Alexandrou1, Nicholas Mifflin2, Craig McManus2,
Vanno Sou3
1Western Sydney University and Liverpool Hospital, Australia
2Liverpool Hospital, Australia
3Campbelltown Hospital, Australia
Introduction One in three hospitalised patients are reported to have non-visible and non-palpable veins, and typically experience numerous painful attempts at peripheral cannulation. Peripheral intravenous catheters (PIVCs) are also known to have high failure rates in patients with difficult access where nearly one in 10 PIVCs fail within eight hours of placement, and nearly half within 24 hours, even if ultrasound is used.
Objectives To increase peripheral catheter dwell by reducing multiple, painful cannulation attempts and complication rates related to peripheral catheters in patients with difficult venous access.
Methods An insertion algorithm was developed to provide the most suitable intravascular device based on ultrasound assessment.
Results Four in every five catheters were inserted first attempt. The insertion algorithm contributed to the majority of devices (n=131, 68%, p<0.001) remaining in place until the cessation of therapy. Median dwell of all devices was 6 days (IQR* 3–11 days). The longest dwell for a peripheral catheter was 69 days. Phlebitis rates were minimal (n=2, 1%, p=0.39) and no infections were reported.
Conclusion Many devices were in dwell for weeks with no complications (totalling 1645 catheter days). Complication rates were significantly lower than has previously been described in the literature for this cohort.
*Interquartile Range
OPTIMISING THE PRODUCT EVALUATION PROCESS
Mrs Joy Blacka1
1VACSA (Vascular Access Consultancy Services Australia), Sydney, Australia
Abstract
This session provided knowledge, examples and measures of success to optimise the product evaluation process. Healthcare facilities, industry and academia depend on clinical experts to undertake effective product evaluation in the clinical setting.
All product evaluations should be treated as a campaign or project and be focused on process improvement. The aim of such projects is to improve patient outcomes through the following:
• improving training and performance
• reducing financial risk
• increasing patient safety
• decreasing hospital-acquired conditions
• creating operational efficiencies.
There are many challenges that present during a product trial and each needs to be proactively managed in a timely fashion. Actively listening and partnering with all stakeholders within the evaluation process, reduce these challenges and enable effective collaboration and a solution-driven culture.
We are here to provide the best care for our patients. Undertaking effective product evaluations is an essential skill for clinical experts, resulting in the uptake of new technologies and improved patient outcomes.
IMPACT OF VARIED MANUAL BOLUS INJECTION FREQUENCY ON PIVC FAILURE, COAGULATION AND TISSUE INJURY – A PRE-CLINICAL HUMAN TRIAL
Samantha Keogh1,2, Alexandra Hawthorn1,2,3, Sapha Mosawy2,4, Annette Billiau5, Lauren Gurney2, Evan Pennell2, Surendran Sabapathy5, Claire Rickard3, A/Prof Andrew Bulmer2,3
1School of Nursing and Institute of Health and Biomedical Innovation and the Alliance for Vascular Access Research and Teaching (AVATAR), Queensland University of Technology, Brisbane, Queensland, Australia
2School of Medical Science and Experimental Laboratory Science (XLabS) Research Group, Griffith University, Gold Coast, Queensland, Australia
3School of Nursing and the Alliance for Vascular Access Research (AVATAR), Griffith University, Brisbane, Queensland, Australia
4Endeavour College of Natural Health, Melbourne, Victoria, Australia
5School of Allied Health Science, Griffith University, Gold Coast, Queensland, Australia
Introduction Peripheral intravenous venous catheters (PIVCs) are associated with a 40-60% post-insertion failure rate. Patient, practice and environmental factors influence failure; however, our understanding of the impact of manual bolus injection (NaCl) on failure is currently inadequate.
Objectives We developed a human, pre-clinical model of bilateral cephalic vein cannulation to study the impact of varied bolus frequency over 5 hours on device failure and vascular structure/function.
Methods Catheters were flushed hourly (high frequency (HF)) or at the beginning and end of the study (i.e., at 0 and 5 hours, low frequency (LF)) with 2x10 mL of sterile saline at each timepoint. The primary outcome was PIVC failure (composite measure). Platelet function, prothrombin time (PT), activated partial thromboplastin time (APTT) and tissue factor (TF) were assessed in the blood. B-mode ultrasound assessed vascular diameter/catheter:vein and thrombus formation over time in a subset. Out of 34 catheters in 17 participants, 1/17 (6%) LF and 0/17 HF catheters failed (p>0.05).
Results Platelet function, APTT and TF were not affected in either condition, or over time. PT increased in HF compared to LF after 5 hours (p<0.05). Venous thromboses occurred in both HF (6/7) versus LF (5/7) conditions (p>0.05). Ultrasound analysis demonstrated similar changes in vascular diameter and blood flow over time, in both groups. PIVC failure was observed in 3% of inserted catheters, however there was no significant difference in HF versus LF devices.
Conclusion HF bolus administration was associated with longer PT times, suggestive of delayed activation or consumption of extrinsic coagulation factors in the absence of TF release.
GLOBAL TRENDS IN PERIPHERAL INTRAVENOUS CATHETER DRESSING, SECUREMENT AND COMPLICATIONS: A SECONDARY ANALYSIS OF 40,637 CATHETERS
Amanda Corley1, Dr. Gillian Ray-Barruel1,2, Claire Rickard1, Amanda Ullman1, Gabor Mihala1,3, Evan Alexandrou1,4
1AVATAR Group, Menzies Health Institute QLD, Griffith University, Brisbane, Australia
2QEII Jubilee Hospital, Brisbane, Australia
3Centre for Applied Health Economics, Menzies Health Institute QLD, Griffith University, Brisbane, Australia
4Department of Intensive Care, Liverpool Hospital, Sydney, Australia
Introduction With over 2 billion peripheral intravenous catheters (PIVCs) used globally each year, the impact of PIVC failure on patients and healthcare organisations is substantial. Effective PIVC dressing and securement may be a key strategy to reduce patient harm. Therefore, an analysis of a global PIVC insertion, management and outcomes dataset, would assist in identifying dressing and securement options effective at reducing PIVC complications.
Objectives To describe global PIVC dressing and securement practices, and any regional variation observed; and, to identify risk factors associated with PIVC site complications and suboptimal dressing and securement.
Methods We performed a secondary analysis of data collected during a cross-sectional study of 40,637 PIVCs in 52 countries. Patient-, PIVC-, and institution-related factors potentially associated with PIVC complications and dressing integrity were extracted from the larger dataset.
Results Transparent polyurethane dressings covered 79% (n=31,968) of PIVCs. Commonly-used methods of PIVC securement were sterile tape around the PIVC hub (n=9,938, 27%) or non-sterile tape over the dressing (n=9,243, 25%). Gauze and tape dressings were associated with reduced insertion site complications (OR 0.58, 95%, CI 0.50-0.68) and better dressing integrity (OR 0.68, 95%, CI 0.59-0.77); whereas, non-sterile tape around the insertion site was associated with increased site complications (OR 2.39, 95%, CI 2.22-2.57) and poorer dressing integrity (OR 1.64, 95%, CI 1.51-1.75).
Conclusion Modifiable risk factors for PIVC complications were identified. Several PIVC dressing and securement combinations were associated with lower rates of complications and are amendable to testing in future randomised controlled trials to address PIVC failure rates.
NO GOING BACK: TUNNELLING CVADS A NURSE LEAD INITIATIVE
Mr Steve Cotterell1
1Canterbury District Health Board, Christchurch, New Zealand
Introduction This presentation outlined advances in nursing skills acquired after attending the WoCoVA Conference in Hong Kong in 2015, and information obtained from the AVAS Conference in Perth 2017.
Objective Following the conferences our objective was to commence a Nurse Lead Practice of Tunnelling Central Venous Access Device’s in patients. We realised there was a need for alternative Central Venous Access Device placement in patients who had problems with long term intravenous access.
Methods We established a Nurse Initiative Credentialing Programme, which was supported by our Interventional Radiologist and our Nurse Consultant Vascular Access.
Results lead to the education of two Interventional Nurses who complete the Credentialing and established the Nurse Lead Service.
Results To date we have successfully inserted 114 Chest Inserted Central Catheter and 57 Tunnelled Peripherally Inserted Central Catheters.
Conclusion Outlined will be the journey from the Conferences to the credentialing of the Advance Nursing Practice in Central Venous Access Device placement.
A SYSTEMATIC REVIEW AND META-ANALYSIS: SAMPLING OF BLOOD FROM PERIPHERAL INTRAVENOUS CANNULA COMPARED WITH VENEPUNCTURE
Dr Linda Coventry1,2, Alycia Jacob1, Hugh Davies1, Laurita Stoneman3, Samantha Keogh4,5, Elisabeth Jacob1
1Edith Cowan University, Joondalup, Australia
2Sir Charles Gairdner Hospital, Nedlands, Australia
3Barinsdale Regional Health Service, Bairnsdale, Australia
4Queensland University of Technology, Brisbane, Australia
5Griffith University, Brisbane, Australia
Introduction Venepuncture can provoke pain, anxiety, and cause varying degrees of trauma to patients. While current guidelines recommend blood samples from peripheral intravenous cannula be taken only on insertion, anecdotal evidence suggests sampling of blood from existing cannulas may be common practice.
Objectives To evaluate if blood samples obtained via peripheral intravenous cannula compared to venepuncture are similar.
Methods A systematic review and meta-analysis was undertaken. Electronic searches were conducted in CINAHL, Cochrane Library, MEDLINE, Scopus, ISI Web of Science, and Joanna Briggs, for English language studies published between January 2000 and April 2017.
The search adhered to the Meta-analysis of Observational Studies in Epidemiology guidelines. The methodological quality of studies was assessed using Joanna Briggs critical appraisal instruments. The overall quality of the evidence was assessed using the GRADE approach.
Results Fourteen studies were identified. Major findings suggest hemolysis rates are higher in blood samples from peripheral intravenous cannula compared with venepuncture; however, hemolysis rates may be lower if a protocol is followed. With regard to equivalence of blood test results, even though some results were outside of the laboratory allowable error and were outside the Bland-Altman Level of Agreement, none of these values would have required clinical intervention. With regard to contamination rates of blood cultures the results were equivocal.
Conclusion Drawing blood samples from peripheral intravenous cannulas may be a viable option for some patient groups when a clear protocol is followed. Further research is required to inform the evidence for best practice recommendations.
PAEDIATRIC AND NEONATAL CENTRAL VENOUS ACCESS DEVICE CONSENSUS PROJECT
Richard Barnes5, Mrs Kerrie Curtis1,2,3, Yves Heloury1, Ed Oakley4,
Amy Shelley1
1Paediatric Integrated Cancer Service, Victoria, Australia
2Alliance for Vascular Access Teaching and Research, Queensland, Australia
3Australian Vascular Access Society, Victoria, Australia
4Royal Children’s Hospital, Victoria, Australia
5Monash Children’s Hospital, Victoria, Australia,
Introduction There is a plethora of evidence for Central Venous Access Device (CVAD) insertion; however, significant gaps exist for the diverse range of maintenance practices. These gaps are often informed by ‘the way we have always done it’, manufacturer recommendations or clinical opinion. The lack of consensus and variation in practices leads to poor vascular access outcomes, especially for the paediatric patient – a high risk and vulnerable patient cohort.
Objective The Lines Project is a collaborative, multidisciplinary, multi-site project that commenced in November 2017 at two tertiary paediatric hospitals in Melbourne, with the aim of implementing evidence-based service improvements and reducing variability in practices, reducing all types of CVAD related complications, and improving patient outcomes.
Method The scope includes all types of CVADs, for preterm to 18-year-olds, across all clinical areas, and the entire CVAD journey, from device selection, insertion and maintenance practices through to removal.
Results The project completed an extensive literature review, thorough scoping of current CVAD management, has reached consensus across the clinical specialties and subsequently is operationalising service improvement recommendations across both health services. The impact of the project is being monitored and evaluated through a newly established CVAD database at each hospital.
Conclusion Variation in CVAD management between clinicians, units and health services increases the risk of poor patient vascular outcomes. Consensus and consistency with current evidence is pivotal to positively impact vascular access outcomes. The Lines Project is making this happen in a paediatric space in Melbourne.
DRESSED FOR SUCCESS!
Mrs Kerrie Curtis1,2,3, Ed Oakley4, Richard Barnes5, Amy Shelly1, Yves Heloury1
1Paediatric Integrated Cancer Service, Victoria, Australia
2Alliance for Vascular Access Teaching and Research, Queensland, Australia
3Australian Vascular Access Society, Victoria, Australia
4Royal Children’s Hospital, Victoria, Australia
5Monash Children’s Hospital, Victoria, Australia
Introduction Dressing management of Central Venous Access Devices (CVADs) requires expert management by the nurse who applies evidence based clinical practice and uses the most appropriate materials according to the needs of the individual patient.
Objective A vascular access project lead by the Paediatric Integrated Cancer Service, Melbourne developed an evidence base education program for CVAD dressing management with the aim of reducing the rate of CVAD dressing related complications such as Medical Adhesive Related Skin Injury (MARSI), catheter migration and accidental dislodgement.
Method The project involves all types of CVADs, in all clinical specialties for all paediatric and neonatal patients in two tertiary paediatric hospitals in Melbourne. Ten key dressing management principles formed the basis of a didactic and practical education program integrating evidence from a literature review and new dressing products into current, dressing-related challenges in clinical practice. The education program was endorsed by the CVAD Committees at both hospitals. The dressing principles are not product or hospital specific so were readily adapted for use by both hospitals.
Results The impact of the education program and clinical practice change is being monitored through a newly established CVAD database at each hospital, which captures the reasons for CVAD removal. Monthly review of the CVAD data will provide information and direction for future education and service improvement opportunities.
Conclusion Consistent application of evidence-based principles has the potential to positively impact the vascular access outcomes of our patients by reducing vascular access complications related to dressing management. A win-win for our patients and healthcare organisations.
COMPUTATIONAL STUDY OF ‘KVO’ IN PERIPHERAL INTRAVENOUS CANNULAS
Dr Barry Doyle1,2, Lachlan Kelsey1,2, Caroline Shelverton3
1Harry Perkins Institute of Medical Research, Perth, Australia
2The University of Western Australia, Perth, Australia
3Flomatrix Pty Ltd, Brisbane, Australia
Introduction Evidence to support an optimum keep-vein-open (KVO) infusion rate for peripheral IV cannulas (PIVC) is lacking.
Objective Our aim was to simulate the fluid dynamics of typical KVOs and better understand the influence of KVO at the PIVC tip.
Method Using computer-aided design, we created a 20G (1.1 mm outer diameter) PIVC and computationally inserted it into two different veins (3.3 and 2.2 mm diameter). Using computational fluid dynamics, we simulated the haemodynamics with a venous flow rate matching the vein diameter and saline infusion rates of 10, 20 and 40 mL/h. We run simulations on 576 cores of a supercomputer (Magnus, Pawsey Supercomputer Centre).
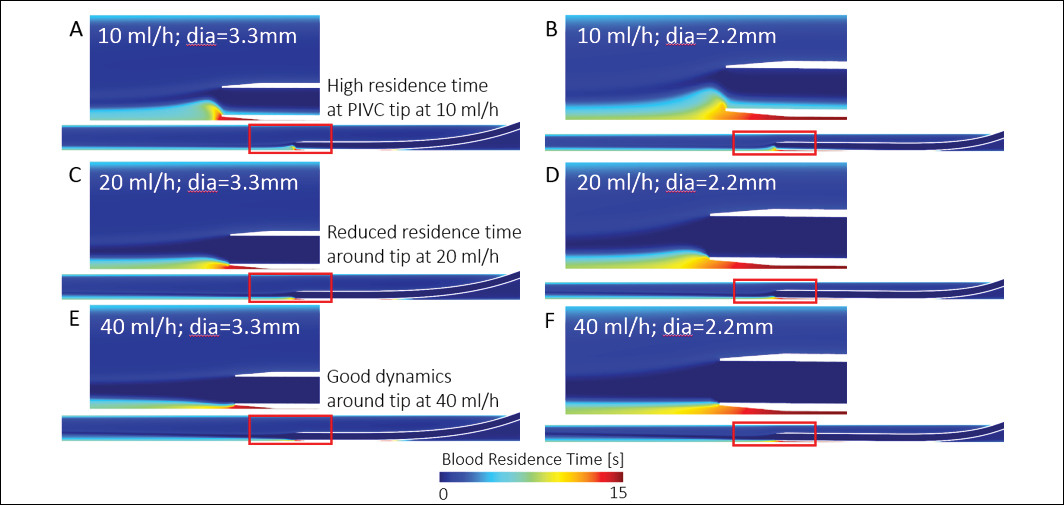
Results At the lowest KVO rate, our data reveals an area of increased blood residence time around the PIVC tip, for both large and small diameter veins (see Figure below). At this low infusion rate, the saline exiting the PIVC is pulled upwards into the faster moving venous blood flow, creating the recirculation zone where RBCs reside. As the KVO increases, this zone diminishes.
Conclusion There are limited data on the different KVO infusion rates and their impact on PIVCs. Here we performed the first computational study on the topic with the hope of providing insight on KVO rates and potential links to thrombus formation and PIVC failure. Although further investigation is needed, our preliminary data suggests low KVO rates may promote clots to occur near the PIVC tip and may influence failure.
PREVENTION OF OCCLUSION OF CENTRAL LINES FOR CHILDREN WITH CANCER: THE POETIC PROJECT
Dr Amanda Ullman1,2, Ms Rachel Edwards1,2, Ms Paula Condon2, Ms Tricia Kleidon1,2, Professor Marie Cooke1, Ms Victoria Gibson1,2, Dr Sonya Osborne3, Professor Claire Rickard1
1Alliance for Vascular Access Teaching and Research; Griffith University, Nathan, Australia
2Queensland Children’s Hospital, South Brisbane, Australia
3University of Southern Queensland, Ipswich, Australia
Introduction Children with cancer require reliable central venous access devices (CVADs); however, one in three central lines used for children with cancer stop working, often because they are blocked.
Objective The aim of this project was to improve early identification and classification of CVAD occlusion and implement standardised evidence-based management strategies
Method A two-phased project was undertaken over 9 months, within the oncology departments at Queensland Children’s Hospital. Phase One involved a prospective audit of CVAD occlusion rates and management practices (n=66 CVADs). Using these data, Phase Two involved the development and implementation of evidence-based strategies to improve CVAD occlusion identification and management, and then repeat prospective audit (n=75 CVADs).
Results During Phase One, 66% of CVADs (n=44) had an occlusive event (as classified by the Catheter Infection and Aspiration System) within the first 21 days (89 per 1000 catheter days), frequently resulting in significant delays to treatment. A large variety of occlusion interventions were used, including frequent position changes, and various flushing techniques, with poor documentation and unsystematic escalation.
During Phase Two, an interdisciplinary group developed a Daily CVAD Patency Assessment, and an Occlusion Management Flowchart, which was implemented via education, mentoring and electronic management system. After implementation, 48% of CVADs (n=36) had an occlusive event within the first 21 days (52 per 1000 catheter days). Overall, systematic escalation of occlusion management increased; however, application of the Occlusion Management Flowchart was variable.
Conclusion CVAD occlusion is common during cancer treatment. Improved prevention, identification and management are essential to ensure early and appropriate treatment.
METHODS FOR MICROBIAL NEEDLELESS CONNECTOR DECONTAMINATION: A SYSTEMATIC REVIEW AND META-ANALYSIS
Ms Julie Flynn1, Professor Samantha Keogh2, Assoc Prof Amanda Ullman1, Ms Emily Larsen1, Prof Claire M Rickard1
1Griffith University, Nathan, Australia
2QUT, Herston, Australia
Introduction Post-insertion management of vascular access devices is essential for the prevention of bloodstream infection. The most commonly used method for needleless connector (NC) decontamination is a 70% alcohol wipe; however, there is little evidence for any method.
Objective The objective of this review was to assess the various products used for NC decontamination to minimise the risk of bloodstream infection in all patient populations, and to evaluate the effectiveness of NC decontamination products to prevent CABSI.
Methodology A systematic search was conducted in CINAHL, Cochrane Central Register of Controlled Trials, Medline and PubMed. The primary outcome was bloodstream infection and included peer reviewed published research, in English.
Results Twelve studies met the inclusion criteria. All the studies where pre-test/post-test design, and concluded that the alcohol wipe was inadequate for NC decontamination. Three studies from each group were included in the meta-analysis. For the chlorhexidine wipe group, the pooled Risk Ratio was 0.40 [95% Confidence interval [CI] 0.18, 0.89]. In the alcohol impregnated cap group, the pooled RR was 0.46 [95% CI 0.23, 0.92].
Conclusion Seventy percent alcohol wipe is commonly used on NCs, prior to healthcare workers accessing the venous system; however, this review suggests that the alcohol wipe is inferior to the 70% alcohol impregnated cap and the CHG wipe. These results need to be taken with caution due to the small sample sizes and pre-test/post-test design. The best method for NC decontamination is not yet established and further high-quality research is needed.
PERIPHERALLY INSERTED CENTRAL CATHETER OUTCOMES POLYURETHANE VERSUS ENDEXO (PIC COMPARE): FEASIBILITY RESULTS OF A PILOT RANDOMISED CONTROL TRIAL
Ms Nicole Gavin1,2, Ms Emily Larsen1,2, Ms Catherine Saxby1, Ms Nicole Marsh1,2, Ms Tricia Kleidon2,3, Associate Professor Amanda Ullman1,2,3, Ms Sarah Northfield1, Mr Gabor Mihala2, Dr Naomi Runnegar4, Professor Claire Rickard1,2,3,4
1Royal Brisbane and Women’s Hospital, Herston, Australia, 2Griffith University, Nathan, Australia, 3Queensland Children’s Hospital, South Brisbane, Australia, 4Princess Alexandra Hospital, Woolloongabba, Australia
Introduction Peripherally inserted central catheters (PICCs) are at risk of failing prior to completion of treatment. Recently, contemporary PICC design incorporating a pressure activated safety valve (PASV®) and new catheter materials, such as Endexo® non-stick polymers, are intended to reduce the adherence of blood components and inhibit thrombus formation thus preventing deep vein thrombosis, occlusion and potentially inhibiting biofilm attachment and subsequent infection.
Objectives Our primary aim was to test the feasibility of conducting a full-scale efficacy randomised control trial (RCT) of a polyurethane PICC without a valve (control) with an Endexo® PASV® PICC (intervention).
Methods A pilot parallel RCT with 1:1 allocation was conducted in a tertiary adult hospital in Brisbane, Australia. The primary outcome was feasibility. Secondary outcomes were PICC failure and complications.
Results One hundred and eleven medical/surgical patients were randomised: 55 in the control and 56 in the intervention. Twelve patients completed 4 weeks of follow-up (5 in the control and 7 in the intervention groups). The inclusion criteria were initially restricted to inpatients, but to achieve recruitment were expanded to include transfers within 24 hours to Hospital in the Home (HITH). Feasibility criteria were met: 86% of eligible patients consented to enrolment and protocol adherence was 93%. Of 83 patients who had their PICC removed due to PICC failure, 23 had not completed their treatment (11 in the control and 12 in the intervention groups).
Conclusion This pilot RCT demonstrated a full-scale efficacy trial is feasible if it includes patients transferred to HITH.
Funding This study received an unrestricted investigator-initiated research grant from Angiodynamics (manufacturer of the intervention PICC). The company had no involvement in study design, execution, analysis or interpretation of results.
SKIN COMPLICATIONS ASSOCIATED WITH PAEDIATRIC CENTRAL VENOUS ACCESS DEVICES: PREVALENCE, INCIDENCE AND RISK
Associate Professor Amanda Ullman1,2,3, Ms Tricia Kleidon1,2, Ms Victoria Gibson1,2, Ms Karen Turner1, Ms Anna Dean1, Ms Paula Cattanach1,2, Ms Colleen Pitt1, Ms Julieta Woosley1,2, Ms Nicole Marsh2,3, Dr Nicole Gavin2,3, Ms Mari Takashima2, Professor Claire Rickard1,2,3
1Vascular Assessment and Management Service, Queensland CHildren’s Hospital, Brisbane, Australia
2Alliance for Vascular Access Teaching and Research (AVATAR), Griffith University, Nathan, Australia
3Nursing and Midwifery Research Centre, Royal Brisbane and Women’s Hospital, Herston, Australia
Introduction Central venous access devices (CVADs) play an important role in the provision of treatment for children with complex health conditions, including trauma, cancer, cystic fibrosis, and other chronic conditions. However, complications affecting the CVAD wound are commonly reported.
Objective The aim of this study was to identify the incidence and prevalence of CVAD-associated skin complications in paediatrics, and describe management, diagnostic and treatment-related characteristics associated with complications.
Method A prospective observational study across medical, oncology and haematology departments at the Queensland Children’s Hospital, Brisbane, Australia, between April and July, 2017. Children with CVADs were assessed twice weekly for CVAD-associated skin complications and associated signs and symptoms. Data was analysed using descriptive statistics (i.e., proportions, frequency) and time-to-event multivariable regression (i.e., hazard ratios).
Results Two hundred and seventy-one CVADs were reviewed over 43,787 catheter days, with less than one in eight participants (14%; n=37) having a CVAD-associated skin complication during their admission (0.95 per 1000 catheter-days [95% CI 0.61-1.17]), most commonly contact dermatitis (11%; n=29; 0.72 per 1000 catheter-days [95% CI 0.50-1.04]). The point prevalence for complications was median 0.4-11% on bi-weekly checks and clinical management was wide-ranging. A primary diagnosis of oncology (HR 2.89 [95% CI 1.10-7.62]) or medical/surgical (HR 2.55, 95% CI 1.04-6.22) conditions, using plain, non-bordered polyurethane dressings (HR 4.92 [95% CI 2.00-12.13]) and poor dressing integrity (HR 2.64 [95% CI 1.18-5.92]) were significantly associated with contact dermatitis.
Conclusion Substantial numbers of paediatric patients experience CVAD-associated skin complications, and strategies to predict, prevent and treat these healthcare-associated injuries are necessary.
THIRTEEN TUNNELLED HAEMODIALYSIS CATHETERS IN TWELVE MONTHS – SURELY TOO MANY: A CASE STUDY
Ms Margaret Golding1
1 Renal Clinical Nurse Specialist. Royal Prince Alfred Hospital, Camperdown, Sydney
Introduction Thrombotic and occlusive complications associated with Vascular Access Devices (VADs) used for haemodialysis is associated with increased morbidity and mortality. Optimising device function is critical to prevent interruption to renal replacement therapy.
Case A 40-year-old female with ulcerative colitis developed interstitial nephritis secondary to sulfarsalazine. In January 2018 she commenced intermittent haemodialysis in a private satellite centre. By August that year she had undergone thirteen re-insertions of her VAD because of thrombotic malfunction and accidental catheter dislodgment. Care predominately consisted of device exchange and warfarinisation. Local policy and procedure limitations at the satellite unit prevented alternative approaches to help maintain and restore catheter patency. A living related renal transplant was performed in January 2019.
Discussion Management of thrombotic occlusive complications related to haemodialysis catheters include various endovascular and pharmacological therapies. Enabling access to tertiary dialysis units may mean more complex and/or expensive evidenced-based treatment strategies can be delivered. This helps to ensure uninterrupted intermittent renal replacement therapy via a VAD until a more permanent solution is achieved. Adequate catheter securement is also of paramount importance to ensure catheter survival.
Pharmacological intervention for catheter-occlusive dysfunction may involve treatment with thrombolytics. After a regimen of a weekly dialysis run at a tertiary dialysis centre (completed with a urokinase intra-luminal lock) and adequate catheter securement, the last VAD remained patent until definitive transplantation occurred.
Conclusion In conclusion, early referral to a tertiary dialysis centre may be required to optimise and maintain catheter function, avoid venous depletion and improve patient outcomes and satisfaction.
DEVELOPMENT OF A PICC NURSE PROCEDURALIST AT THE PRINCE OF WALES HOSPITAL
Ms Jane Holloway1; Mr Mark Sutherland1
1The Prince of Wales Hospital, Barker Street, Randwick, NSW, 2031, Australia
The insertion of peripherally inserted central catheters (PICCs) at The Prince of Wales Hospital started to be performed by registered nurses in the mid-nineties. These nurses were ward-based with vascular access expertise, but they were not vascular access nurses and practice of the procedure was limited by other nursing activities taking priority. By 2011, the demand for PICCs exceeded the service supply of anaesthetist, radiologist and nurses. In 2012, a part-time dedicated PICC insertion service was established by the co-author. The service included a procedure specific assistant and in February 2017, the assistant role was converted into a trainee proceduralist, and training commenced under the mentorship of the co-author.
Theoretical training consisted of PICC care, management and insertion techniques that included ultrasound and electromagnetic/ECG Guidance. Practical training utilised the Bondy Rating Scale of skill development from novice observer to independent proceduralist. The author’s first supervised PICC insertion occurred in May 2017 and by the end of December 2017, the trainee was able to be signed off as an independent proceduralist with a success rate of 81.48% of PICC inserted into the SVC on the first cannulation/pass.
The PICC nurse proceduralist position came about due to the changing demands of modern healthcare and the need for expertise for reliable and effective venous access for the medium to long term with minimal risk-associated complications. Nurses do have the right skill base and knowledge to apply to a proceduralist-based service, and when given the opportunity to be trained it can be applied with success.
RETROSPECTIVE COHORT STUDY OF 1300 PAEDIATRIC CENTRAL VENOUS ACCESS DEVICES: PRACTICE EVOLUTION
Ms Tricia Kleidon, MSc (Nurs Prac)1,2; Professor Claire Rickard, PhD2,3; Ms Jessica Schults, MAppSci (Research)1,2,3; Mr Gabor Mihala, (GCert Biostat)2,4,6; Dr Craig McBride, FRACS2,3,5; Mr John Rudkin7; Dr Brett Chaseling, MBBS (Hons,) FANZCA1; Dr Amanda Ullman, PHD2,3
1Queensland Children’s Hospital, Brisbane, Australia
2Alliance for Vascular Access Teaching and Research (AVATAR), Menzies Heath Institute , Nathan, Australia 3School of Nursing and Midwifery, Griffith University, Nathan, Australia
4Centre for Applied Health Economics, Menzies Health Institute, Nathan, Australia
5Department of Paediatric Surgery, Queensland Children’s Hospital, Brisbane, Australia
6School of Medicine, Griffith University, Brisbane, Australia
7Department of Information and Technology, Queensland Children’s Hospital, Brisbane, Australia
Introduction Insertion of paediatric central venous access devices (CVADs) is challenging, however failure is frequent.
Objective This study aimed to describe practice evolution, complications, and risk factors for multiple insertion attempts and CVAD failure.
Method A retrospective cohort study used prospectively collected data from CVAD database. Data included patient (age, condition, etc.), insertion (indication, device, technique, etc.), and removal (complications, dwell, etc.) characteristics. Descriptive statistics and incidence rates were compared per calendar year. Risk factors for multiple insertion attempts and failure were explored with logistic and Cox models, respectively.
Results Over 273,467 catheter-days, 1,308 CVADs were observed in 863 patients. Ultrasound-guided (USG) insertion increased from 53% (2012) to 58% (2014). Multiple insertion attempts remained static (14%) and significantly associated with non-haematological oncology (odds ratio 2.19; 95% CI 1.08-4.43), respiratory (3.71; 1.10-12.5), gastroenterology (4.18; 1.66-10.5), and other (difficult intravenous access) (2.74; 1.27-5.92). CVAD failure decreased from 35% (2012) to 25% (2014), incidence rate was reduced from 1.50 (95% CI 1.25-1.80) to 1.28 (1.06-1.54) per 1,000 catheter-days. Peripherally inserted CVAD failure was significantly associated with lower body weight (per kilogram, hazard ratio (HR) 1.02; 95% CI 1.00-1.03), cephalic vein (1.62; 1.05-2.62), difficult access (1.92; 1.02-3.73), sub-optimal tip placement (1.69; 1.06-2.69), and gastroenterology diagnosis (2.27; 1.05-4.90). Centrally placed CVAD failure was significantly associated with younger age (per year, HR 1.04; 95% CI 1.00-1.07), tunnelled device (3.38; 2.41-4.73) and gastroenterology diagnosis (1.70; 1.06-2.73).
Conclusion Overall CVAD insertion practices and failure outcomes improved over time. Further improvements and innovation are needed to improve vessel health and preservation for children requiring CVAD.
SMILE – SECURE MY INTRAVENOUS LINE EFFECTIVELY: A PILOT RANDOMISED CONTROLLED TRIAL OF INNOVATIVE PIVC SECUREMENT IN PAEDIATRIC PATIENTS
Ms Tricia Kleidon1,2; Professor Claire Rickard2,3,4; Ms Victoria Gibson1; Mr Gabor Mihala5,6; Hui (Grace) Xu2,5,7; Ms Paula Cattanach1;
Ms Nicole Marsh2,5; Ms Emily Larsen2,5; Assoc Professor Amanda Ullman1,2,5
1Queensland Children’s Hospital, Brisbane, Australia, 2Alliance for Vascular Access Teaching and Research, Brisbane, Australia, 3School of Nursing and Midwifery, Griffith University, , Brisbane, Australia, 4School of Medicine, Griffith University, Brisbane, Australia, 5Nursing & Midwifery Research Centre, Royal Brisbane and Women’s Hospital ,Brisbane, Australia , 6Centre for Applied Health Economics, Menzies Health Institute , Brisbane, Australia, 7Logan Hospital, Queensland Health, Brisbane, Australia
Introduction Peripheral intravenous catheter (PIVC) failure is problematic for clinicians and patients.
Objective This study aimed to test the feasibility of an efficacy randomised control trial (RCT) to determine if new generation dressing and securement products reduce PIVC failure in paediatric inpatients.
Method A three-arm, pilot RCT of 330 participants aged 0-18 years was conducted at Queensland Children’s Hospital, Brisbane. Random assignment to: (i) bordered polyurethane dressing + non-sterile foam (BPU + NSF, standard care); (ii) integrated securement dressing (ISD) + sterile foam; or (iii) BPU + tissue adhesive (TA) + NSF. Primary outcomes were feasibility (eligibility, consent, protocol adherence, retention and acceptability) and PIVC failure (composite of infection, occlusion, infiltration/extravasation and dislodgement). Secondary outcomes were: local/bloodstream infection; occlusion; infiltration; dislodgement; PIVC dwell; safety; patient satisfaction; and dressing adhesion.
Ethics and governance provided through Children’s Health Service, Queensland and prospectively registered on Australian and New Zealand Clinical Trial Registry (ANZCTR) CTRN 12616001073493.
Results Protocol feasibility was achieved, including patient acceptability, timely recruitment, no missing primary outcome data, 96% received allocated intervention, and 0% attrition. PIVC failure occurred: BPU, 47/105, 45% (6.65/1000 hours); ISD, 31/107, 29% (4.40/1000 hours, chi-squared p=0.017) and BPU+TA, 34/107, 32% (5.15/1000 hours, p=0.052). Group differences were non-significant in multi-variable survival analysis (p=0.080 and p=0.283 respectively). Failure risk was associated with younger age (per year, HR 0.93; 0.89-0.97), infection at baseline (HR 1.97; 1.30-2.99) and insertion by non-vascular access specialist (HR 0.39; 0.25-0.62).
Conclusion Both ISD and BPU + TA reduced PIVC failure. A full efficacy RCT to confirm these results is feasible and essential.
HOW OFTEN DO PATIENTS WITH PERIPHERAL INTRAVENOUS CATHETERS EXPERIENCE PRIMARY BLOODSTREAM AND VENOUS INFECTIONS?
Ms Emily Larsen1,2; Professor Claire Rickard1,2,3; Mr Gabor Mihala2,4,5; Ms Nicole Marsh1,2,3; Professor Joan Webster1,2,3; Dr Naomi Runnegar2,6
1Royal Brisbane and Women’s Hospital, Brisbane, Australia
2Alliance for Vascular Access Teaching and Research Group, Menzies Health Institute, Nathan, Australia 3School of Nursing and Midwifery, Griffith University, Nathan, Australia
4School of Medicine, Griffith University, Nathan, Australia
5Centre for Applied Health Economics, Menzies Health Institute, Nathan, Australia
6Princess Alexandra Hospital, Woolloongabba, Australia
Introduction Primary bloodstream infections (BSI) can develop from peripheral venous catheters (PVC), however few studies have evaluated incidence and cumulative risk prospectively, despite the high prevalence of PVC use.
Objective To establish the incidence and daily risk of BSI and local infections associated with PVC, and the impact of dwell time upon outcomes.
Methods Data was synthesised from twelve prospective research studies (11 randomised controlled trials; 1 observational cohort) conducted in Australia (12,942 PVCs, 44,080 device days). The outcome of interest was Primary-BSI, using National Health and Safety Network (2017) criteria. Secondary outcomes were BSI-Laboratory Confirmed BSI (BSI-LCBI (i.e. the PVC is determined as the likely source), local venous infections, and Staphylococcus aureus BSI (SAB). Infections were blind-adjudicated and dwell times were calculated from insertion and removal times.
Results Primary-BSIs occurred in 5/12,942 PVCs (0.04%; or 0.11 per 1000 PVC-days). Of these, there were 3/12,942 (0.02%; 0.07 per 1000 PVC-days) BSI-LCBIs; and one SAB (0.01%; 0.02 per 1,000 PVC-days). Local infections occurred in 4/12,942 PVCs (0.03%). PVCs were in place for <1 to >42 days; Primary-BSIs occurred from Day 2 to Day 7 (Day 1; <1 to 24 hours). No statistically significant difference was found between the lowest daily risk (Day 2, 0.02%, 95%CI 0.00;0.05%) and the highest (Day 7, 0.12%, 95%CI 0.00;0.44%).
Conclusions We identified a low rate of Primary-BSI and local infections under typical Australian hospital conditions; there was no significant increase in BSI risk with dwell time. Ongoing vigilance and prevention strategies remain necessary to prevent Primary-BSI.
HOSPITAL IN THE HOME (HITH) – MANAGEMENT OF PERIPHERALLY INSERTED CENTRAL CATHETERS A RETROSPECTIVE COHORT STUDY
Ms Nicole Marsh1,2,3, Ms Emily Larsen1,2,3, Ms Sam Tapp1, Mr Gabor Mihala3,4,5, Prof Claire Rickard1,2,3
1Royal Brisbane and Women’s Hospital , Herston, Australia
2School of Nursing and Midwifery, Griffith University, Nathan, Australia
3Alliance for Vascular Access Teaching and Research, Griffith University, Nathan, Australia
4School of Medicine, Griffith University, Mount Gravatt, Australia
5Centre for Applied Health Economics, Griffith University, Nathan, Australia
Introduction Worldwide, there has been a shift in methods of healthcare delivery, with an increasing emphasis on avoiding hospital admissions and providing healthcare for patients in their own home. The most common service provided by the Hospital in the Home (HITH) program in Queensland is the delivery of intravenous antibiotics via a peripherally inserted central catheter (PICC).
Objectives Identify prevalence, dwell time, and complications associated with PICCs in the HITH setting.
Methods A single-centre, retrospective cohort study of adults treated by the Royal Brisbane and Women’s HITH team was conducted between 01 June and 15 June 2018. All patients with a PICC were eligible for inclusion. Clinical data was collected by HITH nurses during community visits including: patient characteristics (e.g. age, gender); site assessments (e.g. redness, swelling, tenderness); PICC patency (ability to flush and/or draw blood); and dressing changes. Further information (intravenous medications, medically-diagnosed PICC complications) was collected from medical records.
Results In total, this retrospective study included 164 patients (195 PICCs). The most common reason for HITH admission was a wound infection (n=105; 64%) or bone infection (n=59; 33%). Catheters were often single lumen (n=120; 67%), inserted in the basilic vein (n=137; 80%) by nurses (n=122; 67%). PICC failure occurred in 10% (n= 19); the most common complications were dislodgment (n=9; 5%) and thrombosis (n=4; 2%). There were no confirmed catheter-related blood stream infections.
Conclusion Our study confirms similar PICC failure rates in the HITH community as in hospital inpatients.
A PILOT RANDOMISED CONTROLLED TRIAL OF TWO INSERTION MODELS (EXPERT VERSUS GENERALIST)
Ms Nicole Marsh1,2,3, Prof Joan Webster1,2,4, Ms Emily Larsen1,2,3, Ms Jodie Genzel1,2,3, Prof Marie Cooke1,2,3, Mr Gabor Mihala3,5,6, Prof Claire M Rickard1,2,3
1Royal Brisbane and Women’s Hospital, Herston, Australia
2School of Nursing and Midwifery, Griffith University, Nathan, Australia
3Alliance for Vascular Access Teaching and Research Group, Nathan, Australia
4School of Nursing, Queensland University of Technology, Kelvin Grove, Australia
5School of Medicine, Mount Gravatt, Australia
6Centre for Applied Health Economics, Menzies Health Institute, Nathan, Australia
Introduction It is estimated that two billion peripheral intravenous catheters (PVCs) are sold annually, with post-insertion failure as high as 69%.
Objectives To compare efficacy of expert versus generalist inserter models for successful PVC insertion and subsequent reliable vascular access.
Methods A pilot randomised controlled trial (RCT) of 138 medical/surgical patients was conducted at the Royal Brisbane and Women’s Hospital, comparing PVC insertion by: I) a vascular access specialist (VAS); or, II) any nursing or medical clinician (generalist model). The first primary outcome was feasibility of a larger RCT (e.g., consent, attrition). The second primary outcome was all-cause PVC failure (phlebitis, infiltration/extravasation, occlusion, accidental removal, local or catheter-related bloodstream infection). Secondary outcomes included: dwell time; insertion success; insertion attempts; patient satisfaction; and procedural cost effectiveness.
Results Pilot results demonstrated feasibility of a larger RCT: 92% of screened patients were eligible; there was no attrition or missing outcome data. PVC failure was higher in the generalist group 27/50 (54%) than for VAS 33/69 (48%) (228 vs 217 per 1000 PVC days; Incidence Rate Ratio 1.05, 95% CI 0.61-1.80). All PVCs (n=69) were successfully inserted in the VAS group, compared with 50/69 (72%) successfully inserted in the generalist group. Medical diagnosis and bed-bound status were significantly associated with higher PVC failure in the multi-variable model; securement with additional non-sterile tape was significantly associated with lower PVC failure.
Conclusion This pilot trial confirmed the feasibility and need for a large, multi-centre RCT, comparing VAS with generalist models for PVC insertion.
DRESSING AND SECURING PERIPHERAL INTRAVENOUS CATHETERS: A PILOT RANDOMISED CONTROLLED TRIAL OF A NOVEL INTEGRATED DRESSING
Ms Nicole Marsh1,2,3, Ms Emily Larsen1,3, Ms Jodie Genzel1,3, Mr Gabor Mihala3,4,5, Associate Professor Amanda Ullman1,2,3,6, Ms Tricia Kleidon3,6, Professor Claire M Rickard1,2,3
1Royal Brisbane and Women’s Hospital , Herston, Australia
2School of Nursing and Midwifery, Griffith University, Nathan, Australia
3Alliance for Vascular Access Teaching and Research Group, Nathan, Australia
4School of Medicine, Mount Gravatt, Australia
5Centre for Applied Health Economics, Menzies Health Institute, Nathan, Australia
6Queensland Children’s Hospital, South Brisbane, Australia
Introduction Peripheral intravenous catheter (PIVC) failure remains common. Inadequate stabilisation or securement to the skin, is a known risk factor, as it can allow movement of the catheter within the vein.
Objective To establish the feasibility of a larger randomised controlled trial (RCT) comparing a standard care dressing with a novel dressing and securement method.
Methods A pilot RCT of 300 patients was conducted in the medical/surgical wards of The Royal Brisbane and Women’s Hospital. Eligible patients were randomised to have PIVC dressed with either: (i) a bordered polyurethane dressing (BPU, standard care); or (ii) an integrated securement device (ISD). The first primary outcome was feasibility, including eligibility, consent, protocol adherence and retention rates. The second primary outcome was all-cause PIVC failure: a composite of infection (local or bloodstream), occlusion/infiltration, dislodgement, phlebitis and thrombosis.
Results Feasibility outcomes: 91% of screened patients were eligible; 98% of invited patients consented; 100% of randomised participants received the allocated intervention; and 1/300 (0%) were lost to follow up. PIVC failure occurred in 43/150, 29% (BPU) and 40/150, 27% (ISD) (119 vs 93 per 1000 PIVC days; Incidence Rate Ratio 0.78, 95% CI 0.50-1.23). In the multivariate model, ISD (Hazard Ratio 0.51, 95% CI 0.29-0.89) and admission for surgical emergency was significantly associated with decreased failure. Female gender, wound, hand insertion, and more frequent PIVC use were significantly associated with increased PIVC failure.
Conclusions The multivariable modelling demonstrated that ISD decreased PIVC failure. Feasibility outcomes support the need to undertake a larger trial confirming these results.
LESSONS LEARNT: CREATING A HOSPITAL WIDE VASCULAR ACCESS SERVICE FROM SCRATCH AND DRIVING GROWTH AND EXPANSION IN AN ALREADY ESTABLISHED SERVICE
Tanya Flynn, Oncology Clinical Nurse Consultant1,2,
Anthony Marshall, Central Venous Access Clinical Nurse Consultant2
1St George Hospital, NSW, Australia
2Westmead Hospital, NSW, Australia
This presentation looks at how a dedicated nurse-led team at one Sydney tertiary teaching hospital took a single speciality vascular access service to a whole-of-hospital service; and how a second Sydney tertiary hospital’s nurse-led vascular access team expanded its service to include the insertion of centrally inserted catheters (CVCs), peripherally inserted cannulas (PIVCs), midlines and arterial lines. The presentation is designed to give individuals an idea of the necessary steps to begin the same process of developing a specific vascular access team, and then once established, how to drive growth and broaden skill sets to include the insertion of other vascular access devices in states where it is within their scope of practice.
When clinicians expand their practices in this way, they bring value to their hospitals and themselves by preparing for the inevitable shifts in the healthcare landscape.
As the Australian healthcare system continues to evolve, opportunities have emerged for hospitals to:
• Improve the services they offer patients
• Enhance their reputations as quality care providers
• Reduce the cost of providing care.
Most Australian states already permit nurses to insert CVCs. If hospitals take advantage of this option, their vascular access teams can broaden their services and free up physicians to focus on other tasks and medical procedures.
USING THE PATIENT EXPERIENCE TO IMPROVE IMPLANTED VENOUS PORT ACCESS IN THE EMERGENCY DEPARTMENT (ED) AT BALLARAT HEALTH SERVICE
Miss Robyn Mcintyre, CNE Oncology1, Mrs Fiona Abbott,
CNS ED, 11, Mrs Kerry Missen, RN Div 1 ED,11
1Ballarat Health Service, Ballarat, Australia
Patients receiving chemotherapy for cancer frequently present to the emergency department (ED) after hours when they are unwell or have an elevated temperature. Neutropenic sepsis is often suspected requiring staff in ED to access the patient’s implanted venous port (IVP).
Problems arise when staff do not have the skills or confidence to safely access an IVP, resulting in multiple attempts to establish a peripheral intravenous catheter for treatment. Verbal and formal feedback from patients, carers, and family indicates this practice results in considerable psychological distress. ED staff need to feel safe and competent to access IVPs to ensure optimal treatment outcomes for their patients.
The objective of this education and training package is to deliver a blended program to build the knowledge, skills and attitudes of ED staff towards the safe and competent management of IVPs at Ballarat Health Service.
Adult principles of learning have been applied to develop a program where staff complete a pre-reading package and attend a clinical skills workshop to contextualise key learnings to their work environment.
The workshop has been developed using numerous learning models including VAK (visual, auditory and kinaesthetic) and experiential learning. Modalities include consumer stories (in the form of video vignettes), and simulation on accessing or deaccessing an IVP, blood sampling and troubleshooting.
Two clinical skills workshops have been delivered. A planning meeting has also been completed to engage senior ED staff towards establishing a sustainable model of training in the safe management of all CVADs.
A FEASIBILITY STUDY TO COMPARE CLINICAL PERCEPTION VERSUS THE DIFFICULT INTRAVENOUS ACCESS (DIVA) ASSESSMENT TOOL IN PREDICTING DIFFICULT PAEDIATRIC INTRAVENOUS ACCESS
Mrs Catherine Miller1,2,3; Associate Professor Barbara Parker1;
Doctor Angela Kucia1; Doctor Rebecca Sharp1
1University of South Australia, Adelaide, Australia
2Women’s and Children’s Hospital, North Adelaide, Australia
3SA Medical Imaging
Introduction PIVC insertion is the most common invasive treatment experienced by children. Unsuccessful PIVC insertions increase the risk of physical and psychological adverse effects.
Objectives describe the implementation and validity of method used to test whether the difficult intravenous access assessment tool (DIVA Tool) was superior to clinician assessment in predicting DIVA in children requiring peripherally-inserted intravenous cannula (PIVC) access, determine if level of experience and/or clinical role had any effect on clinicians’ ability to predict DIVA and first-time PIVC insertion success.
Methods In an Australian paediatric medical imaging department, clinicians assessed children prior to attempting PIVC insertion and completed a survey form including prediction of whether the child was likely to have DIVA, rationale, outcome of the attempted PIVC attempt(s), (components of the DIVA Tool were embedded in the survey form, allowing for comparison of the clinician’s assessment and the DIVA Tool prediction score).
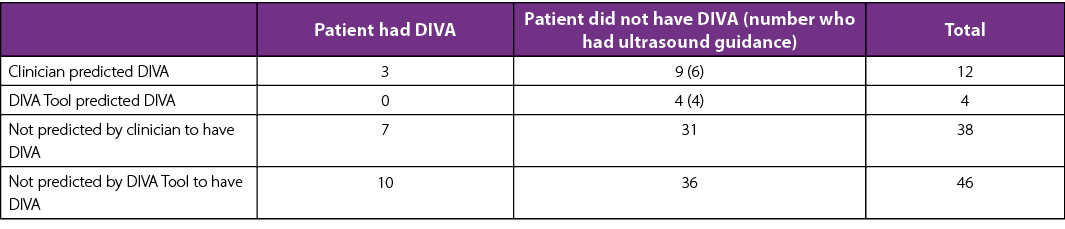
Results 44/50 (88%) of PIVCs were inserted by clinicians with more than five years’ experience, with nurses and doctors having similar success rates. The survey developed was found to be valid. (see table below).
Conclusion The survey developed is valid and could be utilised for future research. The development of a multi-factorial algorithm may be more sensitive than the DIVA Tool. Although ultrasound (US) guided PIVC insertion was successful, previous larger studies have not demonstrated the benefit of US guidance; this warrants further investigation. The similarity between doctors and nurses, in correctly predicting DIVA and successful first-time insertion of PIVCs, supports the development of PIVC insertion nurses.
CENTRAL VENOUS ACCESS DEVICE SECUREMENT AND DRESSING EFFECTIVENESS: THE CASCADE PILOT RANDOMISED CONTROLLED TRIAL IN ADULT INTENSIVE CARE
Prof Marion Mitchell1,2,3, Associate Professor Amanda Ullman1,2, Ms Mari Takashima1,2, Ms Chelsea Davis3, Mr Gabor Mihala1, Ms Madeline Powell3, Ms Victoria Gibson1, Dr Li Zhang1, Ms Michelle Bauer1, Professor E Geoffrey Playford3, Professr Claire M Rickard1,2
1Griffith University, Nathan, Australia
2Princess Alexandra Hospital, Woolloongabba, Australia
3Intensive Care Unit, Princess Alexandra Hospital, Brisbane, Australia
Introduction Central venous access devices (CVADs) are a vital medical device for intensive care (ICU) patients, however CVAD complications and failure are common, yet potentially prevented through effective dressings and securement.
Objectives To test the feasibility of a randomised controlled trial (RCT) comparing standard care with three dressing and securement products, to prevent CVAD failure, in ICU participants. Secondary aims included comparing dressing and securement products on CVAD failure, microbial colonisation and intervention costs.
Methods A single-centre pilot RCT of ICU adult patients requiring CVADs for >24 hours were randomised to: (i) sutures plus chlorhexidine gluconate (CHG) dressing (Standard-care-group); (ii) standard-care plus tissue adhesive (TA-group); (iii) two sutureless stabilisation devices plus CHG dressing (SSD- group); and (iv) sutures, CHG disc plus integrated securement dressing (ISD-group). Descriptive statistics were used to report feasibility, and baseline comparisons. Incidence rates (IR) of CVAD failure were calculated with Fisher’s exact and log-rank tests. Cox regression explored baseline risk for failure.
Results A total of 121 participants were randomised. Study feasibility was established with no withdrawal or attrition, and high staff acceptability of study interventions. Catheter failure was highest in SSD group (IR 27.3/1000 catheter days [95% CI 11.4-65.6]) and lowest in the ISD group (IR 8.8/1,000 catheter days [95% CI 2.19-35.0]. Most complications were suspected central line-associated bloodstream infections; however, only one was laboratory confirmed (Standard-care-group). Costs were lowest in the Standard-care-group.
Conclusion The three innovative treatment arms are feasible interventions to be tested in a full-scale RCT, to inform measures to prevent CVAD failures in ICU patients.
DEVELOPMENT OF AN ALGORITHM USING ULTRASONOGRAPHY-ASSISTED PERIPHERAL INTRAVENOUS CATHETER PLACEMENT FOR REDUCING CATHETER FAILURE: UNCONTROLLED BEFORE AND AFTER STUDY
Associate Professor Ryoko Murayama1,2; Graduate student Chiho Kanno3; Assistant Professor Mari Abe1; Assistant Professor Toshiaki Takahashi4; Graduate student Yui Shintani3; Head Nurse Junko Nogami5; Director of Nursing, Chieko Komiyama5; Professor Hiromi Sanada2,3
1Department of Advanced Nursing Technology, Graduate School of Medicine, The University of Tokyo, Hongo, Bunkyo-ku, Japan
2Global Nursing Research Center, Graduate School of Medicine, The University of Tokyo, Hongo, Bunkyo-ku, Japan
3Department of Gerontological Nursing/Wound Care Management, Graduate School of Medicine, The University of Tokyo, Hongo, Bunkyo-ku, Japan
4Department of Life Support Technology (Molten), Graduate School of Medicine, The University of Tokyo, Hongo, Bunkyo-ku, Japan
5Department of Nursing, The University of Tokyo Hospital, Hongo, Bunkyo-ku, Japan
Abstract
Up to 50% peripheral intravenous catheters (PIVs) are removed prematurely because of failures. Catheter failure (CF) leads to replacement and is a great concern for patients and medical staff. It is known that visualization of catheters and vessels with ultrasonography (US) during placement prevents CF. However, US is not a common technique for general nurses in Japan. In order to standardize US-assisted PIV placement techniques, an algorithm is needed. This study aimed to develop an algorithm using US-assisted PIV placement to reduce CF rate. Furthermore, to evaluate the effectiveness of the algorithm, CF rates were compared before and after intervention. An uncontrolled before and after study was performed. The intervention was PIV placement using the algorithm by 23 nurses undergoing training. Intention to treat, per protocol analyses, was applied. Logistic regression analysis was used for factor analysis. This study was approved by the Research Ethics Committee of the University of Tokyo (No.11832-(3)). The CF rate in the before-intervention group 35.2% (19/54) did not significantly differ from after intervention group 33.6% (48/143; p=.831), yet significantly differed from complete algorithm-use group 8.7% (2/23; p=.017). In factor analysis, compliance to the algorithm was significantly correlated with CF (p=.032). The compliance rate was low 16.1% (23/143). Algorithm compliance reduced CF by confirming appropriate catheter tip position from the insertion to the securement phase. This algorithm effectively reduced CF, however, the compliance rate was unacceptable. In order to increase the compliance rate, modified training tools and new visualizing technology is required.
A NOVEL HEALTHCARE LITERACY INITIATIVE FOR PERMANENTLY AND SUSTAINABLY UPGRADING INFECTION PREVENTION AND VASCULAR ACCESS BEST PRACTICE
Mr Ramzy Nasrallah1
1Association for Vascular Access, Flemington, United States
Germ theory was first proposed in 1546. Three centuries later, hand hygiene was found to be an effective method for preventing communicable disease. We have known how germs are spread – and how to reduce transmission – for hundreds of years.
Vascular access is healthcare’s most common invasive procedure. Hundreds of millions of intravascular devices are placed in patients each year. Dozens of patients die each day from catheter-related complications and infections.
Hand hygiene and vascular access are two ubiquitous and vital elements of healthcare delivery.
Thousands of patients are discharged from hospitals every day with implanted vascular access devices. Many are readmitted with expensive and sometimes fatal complications resulting from improper non-acute care.
Treating these preventable mistakes costs global healthcare systems billions of dollars every year.
The Association for Vascular Access (AVA) has developed a novel healthcare literacy program called Vicarium. It is a customized healthcare strategy that activates the patient to support hospital policies and outcome initiatives – from admission to discharge, and throughout the continuum of care.
Vicarium:
- Aligns patient expectations for hand hygiene and vascular access to hospital policies upon admission.
- Creates a portable standard of care/maintenance and patient expectations that extends beyond the hospital throughout an often fragmented non-acute care continuum
- Develops a channel into hospitals for the latest vascular access research, clinical education and resources
- is a permanent, scalable, and sustainable literacy program designed to upgrade infection prevention and vascular access compliance from the invitation of therapy through completion, regardless of setting
A NEXT-GENERATION ULTRASOUND-GUIDED CONFIRMATION SYSTEM TO IMPROVE PERIPHERALLY INSERTED CENTRAL CATHETER (PICC) INSERTION SUCCESS AND OPTIMAL PLACEMENT: A QUALITY IMPROVEMENT ACTIVITY
Ms Emily Larsen1,2,3, Ms Megan Nicholas1, Ms Nicole Marsh1,2,3, Mr Gabor Mihala3,4,5, Mr Peter Groom1, Prof Claire M Rickard1,2,3
1Royal Brisbane and Women’s Hospital, Queensland, Australia
2School of Nursing and Midwifery, Griffith University, Queensland, Australia
3Alliance for Vascular Access Teaching and Research Group, Menzies Health Institute, Queensland, Australia
4School of Medicine, Griffith University, Queensland, Australia
5Centre for Applied Health Economics, Menzies Health Institute, Queensland, Australia
Introduction Peripherally inserted central catheters (PICCs) are commonly placed for the administration of intravenous fluids, medications, chemotherapy and parenteral nutrition. Innovative ultrasound (US) technologies used to assist PICC inserters continue to emerge; however, the impact of such technologies upon clinical outcomes is not well understood.
Objective This quality improvement (QI) activity aimed to: (i) evaluate the incidence of successful PICC insertion (optimal tip positioning) after introduction of a next-generation tip confirmation-enabled USS system; and (ii) further assess the impact upon incidence of PICC failure.
Method A pre-post retrospective cohort study of adult patients requiring a new PICC was conducted within a nurse-led insertion service of a large Brisbane tertiary hospital. Data collected between 01 May 2017 (4-months pre- equipment introduction) and 31 January 2018 (4-months post-equipment) was included, with a 4-week ‘wash-in’ education/learning period (September) excluded. Data including patient characteristics (e.g., gender, age), PICC characteristics (e.g., size, location) and device removal details were collected and entered prospectively by insertion staff.
Results In total, n=476 PICCs were included in the analysis. Incidence of insertion failure was similar between the intervention (post-) group (n=226, 8%) and the control (pre-) group (n=250, 7%) (p=0.76). Similarly, there was no statistically significant difference in the incidence of PICC failure between in the intervention (post-) group (21%), compared with the control (pre-) group (18%) (p=0.53).
Conclusion The QI activity demonstrated no immediate improvement upon clinical outcomes with the use of next-generation US technology; however, anecdotal feedback suggests technology features (e.g., catheter-to-vein ratio) carry benefits.
IMPLEMENTING A HOSPITAL-WIDE POINT-OF-CARE ULTRASOUND SKILLS TRAINING AND ACCREDITATION PROGRAM: PHASE ONE PERIPHERAL USG IV ACCESS
Dr Elizabeth Prentice1, Dr Adam O Brien
1Royal Children’s Hospital Melbourne, Melbourne, Australia
Point-of-care ultrasound (POCUS) has slowly made its way into clinical practice as an essential aid to many point-of-care applications.
Much benefit to patient care can be achieved with the use of POCUS, but unfortunately harm, missed diagnoses, damaged equipment, and breached sterility can also occur if used incorrectly.
Some professional colleges have embraced this with compulsory accreditation, others are still processing how this can be achieved. Hospitals need to provide safe systems for accreditation in and use of point-of-care-ultrasound for staff and patients.
This ambitious project was approved by Royal Children’s Hospital Melbourne – to ensure all users are accredited, they are exposed to a standard didactic training program, skills training, up-to-date information, equipment, and then formal skills maintenance.
Phase one has been implemented and will be presented – formal training and accreditation of multiple nursing, medical and technician practitioners in ultrasound-guided peripheral IV access, purchase of multiple ultrasound units.
The challenge of how many to train and how ward patients outside critical care areas get access to ultrasound guided IV access without a formal peripheral IV access team, is still a work in progress
PIVC INSERTION AND MANAGEMENT: TO BUNDLE OR NOT TO BUNDLE? THAT IS THE QUESTION!
Dr Gillian Ray-Barruel1,2,4,5, Ms Hui Grace Xu1,3, Ms Nicole Marsh1,4, Prof Marie Cooke1, Prof Claire Rickard1,4,5
1Griffith University School of Nursing and Midwifery, Nathan, Australia
2QEII Jubilee Hospital, Coopers Plains, Australia
3Logan Hospital, Meadowbrook, Australia
4Royal Brisbane and Women’s Hospital, Herston, Australia
5Princess Alexandra Hospital, Woolloongabba, Australia
Introduction Following reported success of central line bundles for preventing bloodstream infection, many hospitals are considering implementing a peripheral intravenous catheter insertion or maintenance bundle. The effectiveness of PIVC bundles is unclear.
Objectives To identify if implementation of PIVC insertion and/or maintenance bundles can prevent bloodstream infection in hospital patients.
Methods Systematic review. We searched electronic databases PubMed, CINAHL, EMBASE, Medline, Cochrane CENTRAL, ISI Web of Science, trial registries, and grey literature for eligible studies. Inclusion criteria: intervention studies evaluating PIVC insertion or maintenance bundles with two or more components, published in English (2000–2018). Search terms included: peripheral intravenous catheter/cannula, insertion, maintenance, bundle, infection, infiltration, extravasation, dislodgement, thrombosis, occlusion and phlebitis. Two reviewers independently conducted data extraction and quality assessments using the Downs and Black checklist.
Results We screened 14,458 records, and 13 studies (6 interrupted-time series, 7 before-and-after) met the inclusion criteria. Both insertion and maintenance bundles included multiple components (2–7 items per bundle) and most were not based on evidence. Despite implementing different bundles, most studies reported reductions in phlebitis and bloodstream infection. Methodological quality of all studies ranked between ‘low’ and ‘fair’.
Conclusions We found a wide range of insertion and maintenance PIVC bundle items and most were not based on evidence. A lack of consistency in bundle components and diverse implementation strategies precludes meaningful comparison at this time. More robust research is needed to determine the effect of PIVC bundles on preventing complications and bloodstream infection.
A PROCEDURE FOR INSERTION AND MAINTENANCE CARE OF INTRA-ARTERIAL CATHETERS USING BUNDLED INTERVENTIONS
Dr Heather Reynolds, RN, PhD1,3; Dr John Gowardman, MBChB (Auck), FCICM, FRACP2,3
1Royal Brisbane Hospital, Brisbane, Australia
2The University of Queensland, Brisbane, Australia
3Women’s Hospital, Brisbane, Australia
Introduction Intra-arterial catheters do not command the same attention in the literature as central venous catheters in spite of their widespread use. Importantly, their risk of infection is comparable with short-term, non-cuffed central venous catheters.
Objective To implement a hospital procedure incorporating the bundle concept for insertion/maintenance care of peripherally inserted intra-arterial catheters to minimize infection risk.
Method An arterial catheter procedural document was developed using Provonost’s concept of several evidence-based procedures to reduce catheter-related bloodstream infections. This procedure for adult patients provided best practice standards which incorporated:
• A process to support safe insertion/management with 5 bundle components
(i) Hand Hygiene
(ii) Aseptic Non-Touch Technique (ANTT)
(iii) Skin Preparation
(iv) Dressing
(v) Documentation;
• Guidance regarding assessment/site monitoring and device selection; and
• Instruction for safe blood sampling/accurate specimen collection.
Results The procedure for insertion/maintenance of intra-arterial catheters was approved by Quality and Safety, and has been published as a procedure for management of intra-arterial catheters in the departments of emergency medicine, operating theatres, post-anaesthetic care and intensive care medicine at Royal Brisbane and Women’s Hospital. Insertion/dressing trolleys containing all components/checklists were located at easy access points, facilitating implementation. An education program for staff from emergency, anaesthetic/perioperative and intensive care addressed infection prevention strategies.
Conclusions A positive impact after introducing a new procedural document relies on timely implementation and learning from facilitators/barriers. Education sessions and communication by Departmental Directors have overcome many challenges in our healthcare facility. Future analysis of infectious complications and staff attitudes will assess the procedure/bundle’s effectiveness and outcomes.
VASCULAR ACCESS OUTCOMES REPORTED IN PAEDIATRIC TRIALS: THE NEED FOR A STANDARDISED OUTCOME SET
Ms Jessica Schults1,2,3; Professor Claire M Rickard1,;, Ms Tricia Kleidon1,2,;, Ms Rebecca Hughes;, Dr Fiona Macfarlane3; Mr Jacky Hung4; Associate Professor Amanda Ullman2,3
1Alliance for Vascular Access Teaching and Research (AVATAR) Group, Menzies Heath Institute Queensland, Griffith University, Queensland, Australia
2School of Nursing and Midwifery, Griffith University, Nathan, Queensland, Australia
3Department of Anaesthesia and Pain Management, Queensland Children’s Hospital, South Brisbane, Queensland, Australia
4Centre for Children’s Health Research, South Brisbane, Queensland, Australia
Introduction Approximately 70% of hospitalised children require a vascular access (VA) device. In paediatrics, a standardised outcome set to be reported as a minimum in VA trials does not exist. Subsequently, it is not known what outcomes researchers choose and whether these are important to key stakeholders, mainly patients, clinicians and policy makers.
Objective To describe the range and heterogeneity of outcome measures reported in paediatric VA trials.
Method Scoping review framework. A systematic search for randomised controlled trials (RCTs) reporting VA outcomes in paediatrics was undertaken in the Cochrane library, EMBASE, CINAHL, PubMed, MEDLINE and EBSCO using medical subject headings and key words related to VA and paediatrics. We included RCTs of children (0-18 years) reporting any VA outcome. The frequency and definition of outcome measures were extracted and evaluated.
Results The review identified 93 trials, from 32 countries, reporting 214 outcome measures. The most commonly reported outcome domains were insertion (44 RCTs; 47%; 69 outcome measures), non-infectious complications (33 RCTs; 35%; 35 outcome measures), and infectious complications (30 RCTs; 32%; 33 outcome measures). Patient reported outcomes were infrequently reported (21 RCTs; 22%; 14 outcome measures).
Conclusion There is significant multiplicity of outcomes and heterogeneity among outcome measurements in paediatric VA trials. Further work needs to be undertaken to establish a consensus-based set of outcomes to be reported as a minimum in all VA trials. This will facilitate data comparison to benchmark outcomes, evaluate quality initiatives, and improve practice.
DIFFICULT INTRAVENOUS ACCESS IN CHILDREN: AN INTERNATIONAL PRACTICE SURVEY
Ms Jessica Schults1,2,3 Professor Claire M Rickard1,2, Ms Tricia Kleidon1,2,3, Ms Rebecca Paterson2, Dr Fiona Macfarlane3, Associate Professor Amanda J Ullman1,2
1Alliance for Vascular Access Teaching and Research (AVATAR) Group, Menzies Heath Institute Queensland, Griffith University, Queensland, Australia
2School of Nursing and Midwifery, Griffith University, Nathan, Queensland, Australia
3Department of Anaesthesia and Pain Management, Queensland Children’s Hospital, South Brisbane, Queensland, Australia
Introduction Peripheral venous cannulation is considered a routine procedure, yet 50% of first attempt insertions fail, necessitating repeat needle-sticks. The early identification and prompt escalation of children with difficult intravenous access (DIVA) can improve first attempt success; however, it is not known what clinical resources are available to guide clinicians’ practice.
Objective To describe current international practice regarding the identification and management of children with DIVA.
Method A cross-sectional, international survey; followed by a systematic review and critical appraisal of clinical pathways using the AGREE II checklist.
Results A total of 148 clinicians from eight countries completed the survey. The majority were nurses (92; 62%), practising as vascular access specialists (27; 18%). Twenty-three (16%) respondents reported using a DIVA tool, of which the ‘DIVA Score’ was most common (5; 22%). Five clinical pathways were identified from the survey and review. Based on the AGREE II domains, pathways generally scored well for scope and purpose, and clarity of presentation areas. Information on the rigour of development and editorial independence were infrequently detailed. Based on AGREE II findings, one pathway was recommended for clinical practice, and four were recommended for use with modification.
Conclusions Resources for the identification and management of children with DIVA are not standardised or consistently used. Further work is needed to streamline processes for DIVA identification and escalation to an appropriate clinician who has competence in technology assisted insertion. This will enhance patient experiences and reduce harm from multiple insertion attempts.
NEEDLELESS CONNECTOR CARE: AN OBSERVATIONAL STUDY
Ms Karen Slater1,2; Professor Marie Cooke3,4; Ms Emma Scanlan1; Professor Claire M Rickard3,4
1 Princess Alexandra Hospital, Queensland, Australia
2 School of Nursing and Midwifery, Griffith University, Queensland, Australia
3 Alliance for Vascular Access Teaching & Research (AVATAR) Group, Menzies Health Institute Queensland, 4School of Nursing and Midwifery, Griffith University, Queensland, Australia
Introduction The majority of hospital patients have a peripheral intravenous catheter (PIVC) at some stage during their hospitalisation. Good hand hygiene and “scrub the hub” practices decrease the possibility of patients’ developing a blood stream infection (BSI). Patient BSIs lead to increased morbidity, mortality and higher costs.
Objective The aim of the study was to observe nurses’ hand hygiene and medication administration practices associated with needleless connectors (NC) attached to PIVCs.
Method This observational study was conducted in two medical and surgical wards, and the emergency department of a tertiary hospital. Observations were undertaken using a standardised collection tool. The same Clinical Nurse with expertise in vascular access completed all observations. Hand hygiene compliance was observed pre-medication preparation, pre-administration and post-procedure. Length of NC decontamination and drying time was also measured.
Results High rates of compliance with NC decontamination (99%) were observed, but the average scrub time was only 6.1%. Hand hygiene prior to medication preparation was 33%, immediately prior to medication administration and NC access was 43%, and 65% post medication administration. The Emergency Department setting, and glove use were associated with the poorest hand hygiene compliance (p<0.01).
Conclusion This study observed high rates of NC decontamination, but with a shorter duration of “scrub the hub” than recommended by International Guidelines. Hand hygiene rates both prior to medication preparation and prior to accessing the NC were poor, considerably lower than the Hand Hygiene Australian target.
HOW LONG DOES IT TAKE NEEDLELESS CONNECTORS TO DRY AFTER SCRUBBING?
Ms Karen Slater1,2, Ms Fiona Fullerton1, Prof Marie Cooke3,4,
Ms Simone Snell1, Prof Claire M Rickard3,4
1Princess Alexandra Hospital, Brisbane, Australia
2School of Nursing and Midwifery, Griffith Univsersity, Brisbane, Australia
3Alliance for Vascular Access Taching & Reserch (AVATAR) Group
4School of Nursing and Midwifery, Griffith University, Brisbane, Australia
Introduction It has long been established that needleless connectors (NCs) need to dry after decontamination prior to them being accessed. There is little evidence as to how long they take to dry and if different antiseptics dry at different times.
Objective The objective of this study was to establish NC drying time for standard swab preparations for 70% isopropyl alcohol, 70% isopropyl alcohol and 2% chlorhexidine (CHG), and 10% povidone-iodine.
Method The study was conducted in an air-conditioned hospital meeting room, with a research assistant simulating a patient. The NC was decontaminated for 15 seconds. It was held at the base and allowed to dry. The NC was considered dry when moisture could no longer be observed on tissue paper. Observations for dryness commenced at 15 seconds, and were repeated at 5-second increases or decreases until dryness was established. Each test required a new 15 second scrub. For povidone-iodine this was increased to 30-second intervals. Once dry was established the procedure was repeated four times.
Results After a 15-second scrub, 70% isopropyl alcohol was dry at 5 seconds, 70% isopropyl alcohol and 2% CHG was dry at 20 seconds, 10% povidone-iodine remained wet at 6 minutes.
Conclusion Antiseptic type significantly affects NC drying time in the context of similar fluid volume and pad/wipe material with a standard NC decontamination time. The 70% isopropyl alcohol appeared most efficient in terms of dry time, with povidone-iodine taking >6 minutes making its recommendation impractical for us.
A SYSTEMATIC REVIEW OF RECENT TRIALS EVALUATING THE IMPACT OF CENTRAL VENOUS CATHETER DESIGN AND MATERIALS ON THROMBOSIS
Mr Eugene Slaughter1, Dr Kathryn Kynoch2, Miss Megan Brodribb3, Professor Samantha Keogh1
1QUT School of Nursing and Institute of Health and Biomedical Innovation, Kelvin Grove, Brisbane, Australia
2Evidence in Practice Unit, Mater Health, Brisbane, Australia
3Division of Administrative Services, QUT Library, Brisbane, Australia
Introduction Thrombosis is a common complication associated with central venous catheter insertion.
Several anti-thrombogenic materials and alterations to catheter design have been developed to lower thrombosis rates.
Methods A systematic search was conducted in CINAHL, Embase, Medline and PubMed. Trial registries and grey literature was also searched. The primary outcome was catheter-related thrombosis, with only randomised controlled trials included. Inclusion criteria: English language, any age group, within last 10 years, reporting impact of different catheter designs and/or materials on thrombosis. Quality assessment was performed using the Cochrane Rik of Bias Tool. A narrative synthesis and meta-analysis were conducted.
Results From a possible 192 studies, eight studies met the inclusion criteria and were assessed for inclusion. Three studies (n=1275) investigated different catheter materials; four studies (n=597) compared different catheter designs, and one study (n=150) evaluated impact of combined design and material on outcomes relevant to this review. Meta-analysis demonstrated that neither catheter material nor design alone or in combination had a significant impact on thrombosis (RR: 0.94 [95% CI 0.83, 1.05]). Different catheter materials and design also had no significant impact on occlusion or catheter-related bloodstream infection. Studies were of mixed quality overall.
Conclusion Different central venous catheter materials and/or design were not associated with a reduction in the risk of either catheter related thrombosis or infection. However, overall reporting and small sample sizes make it difficult to draw firm conclusions. Larger, randomised studies are required to provide confirmatory evidence about the possible merits of innovative catheter design on patient outcomes.
HOW MANY IS TOO MANY? DEVELOPMENT OF AN ASSESSMENT TOOL TO IMPROVE PATIENT OUTCOMES
Ms Penelope Stevens, Clinical Practice Coordinatior1; Mrs Julie Evans, Nurse Unit Manager1
1Cancer Care Service, Wide Bay Hospital Health Service, Hervey Bay, Australia
Introduction Peripheral intravenous cannulation is the most common invasive procedure in clinical practice. Around 60% of adults are difficult to cannulate. In 2016 approximately 2.5 million cannulas were used in Queensland public hospitals, it is reported that 50% of them failed. It has been shown that careful planning and assessment of a patients’ vascular anatomy and identifying their comorbidities will impact on the success rate of cannulation.
Objective Our current hospital policy states two attempts per clinician, it does not say how many clinicians can attempt the insertion. We have no escalation policy for failed cannulation, we have no tool. The purpose of our project was to develop an assessment tool and change current practice.
Method An audit of 60 electronic records of patients attending the chemotherapy unit identified up to eight failed attempts of cannulation per episode of care. Benchmarking against tertiary hospitals identified that some had escalation policies in place to overcome repeated fails. Recommendations from leading vascular societies promote the use of a pre-cannulation assessment.
Results This has led us to develop a tool that can be used whenever a patient requires cannulation. This tool prompts the inserter to consider patients’ chronic conditions and their vascular anatomy. It also takes into consideration past failed attempts, patient preference, and the complexity of comorbidities that many of our ageing population in the Wide Bay experience.
Conclusion When we plan for vascular access, we can reduce unwanted complications, prevent delays in treatment, and ultimately improve patient comfort and satisfaction.
WHY DID WE GET HERE? ULTRASOUND AND ELECTRO-MAGNETIC/ECG GUIDANCE AT THE PRINCE OF WALES HOSPITAL: A PERSONAL EXPERIENCE
Mr Mark Sutherland, Clinical Nurse Consultant, Enteral and Parenteral Therapy1
1The Prince of Wales Hospital, Randwick, NSW, Australia
Abstract
The peripherally inserted central catheter (PICC) revolution started at The Prince of Wales Hospital in the mid-nineties with the introduction of hospital-in-the-home (HITH) programs and improved device technology. The PICC was identified as suitable, because it was perceived as being low risk, capable of safe bedside insertion, and equal dwell time to treatment ratio. With time, PICC’s issues and short comings evolved and needed to be addressed.
Prior to the availability of ultrasound, electromagnetic (EM) and ECG guidance, PICC insertion could be a frustrating procedure. Difficulties were encountered achieving successful catheter insertion or ideal tip position. For example, in 2002, 84 PICC insertions were attempted by the author, only 56 (66.67%) of the distal tips were able to be inserted into the superior vena cava (SVC) as required for reliable catheter performance and thrombosis avoidance.
Needed was a procedural evolution to assist in obtaining optimal site selection and PICC distal tip placement. In 2009, the author obtained the access of ultrasound and in 2014 introduced EM/ECG guidance. In 2018, the author and his assistant attended 260 PICC insertion procedures, 253 (97.31%) distal tips were successfully inserted into the SVC.
Ultrasound allows the proceduralist to no longer be restricted to the antecubital fossa. It gives the proceduralist the ability to assess catheter to vessel ratios, offer more access choices, and visually control venepuncture. EM and ECG guidance offers the accuracy of perfectly placed catheter distal tips that previously had only been available to catheters inserted under fluoroscopy.
CATHETER TIPS ARE POSSIBLE RESOURCES FOR BIOLOGICAL STUDY ON CATHETER FAILURE
Dr. Toshiaki Takahashi1, Dr. Takeo Minematsu2,3, Dr. Ryoko Murayama3,4, Prof. Taketoshi Mori1,3, Prof. Hiromi Sanada3,5
1Department of Life Support Technology (Molten), Graduate School of Medicine, the University of Tokyo, Tokyo, Japan
2Department of Skin Care Science, Graduate School of Medicine, the University of Tokyo, Tokyo, Japan
3Global Nursing Research Center, Graduate School of Medicine, the University of Tokyo, Tokyo, Japan
4Department of Advanced Nursing Technology, Graduate School of Medicine, the University of Tokyo, Tokyo, Japan
5Department of Gerontological Nursing/Wound Care Management, Graduate School of Medicine, The University of Tokyo, Tokyo, Japan
Introduction Peripheral intravenous catheter failure (CF) is an important clinical problem. Our previous study revealed the presence of thrombus and subcutaneous edema around the tips of failed catheters, suggesting the contribution of these phenomena to CF. Although further study of the molecular mechanisms is required for a better understanding of CF, such investigation has been overlooked.
Objective In this study, we performed histological and molecular biological analyses of the catheter tip to demonstrate its potential as a resource for biological investigation.
Method The CF (+) group included 30 failed catheters involving thrombus and subcutaneous edema identified by ultrasonography. The CF (-) group included 26 catheters completed normally. The removed catheter tips were fixed for hematoxylin-eosin (HE) staining and real-time reverse transcriptase polymerase chain reaction was performed for eukaryotic 18S ribosomal RNA (rRNA), interleukin (IL)-1β as leukocytic pyrogen, tumor necrosis factor (TNF)-α, tissue plasminogen activator (tPA), and plasminogen activator inhibitor (PAI)-1.
Results HE staining identified attached nuclear cells on the inner surfaces even in the control catheter. The 18S rRNA was amplified in all samples. The expression level of PAI-1, an inhibitor of thrombolysis, was significantly higher in the CF (+) group than in the CF (-) group (p=0.009), whereas the expression levels of other genes did not differ between the groups.
Conclusion These results suggested that catheter tips are a potential resource for biological investigation, and expression analysis of the attached cells can reflect pathological conditions of the catheterized tissue. Further studies using catheter tips are required to elucidate the molecular mechanisms of CF.
WOUNDED: A SECONDARY ANALYSIS EXPLORING SKIN COMPLICATIONS ASSOCIATED WITH VASCULAR ACCESS DEVICES
Dr Amanda Ullman, Associate Professor1,2,3; Mr Gabor Mihala1,4; Ms Kate O’Leary1; Ms Nicole Marsh1,2; Ms Christine Woods1,;, Dr Simon Bugden5; Dr Mark Scott5; Professor Claire Rickard1,2,3
1Alliance for Vascular Access Teaching and Research (AVATAR); Griffith University, Nathan, Australia
2Royal Brisbane and Women’s Hospital, Herston, Australia
3Queensland Children’s Hospital, South Brisbane, Australia
4Centre for Applied Health Economics, Griffith University, Nathan, Australia
5Caboolture Hospital, Caboolture, Australia
Introduction Insertion of vascular access devices creates a wound, vulnerable to irritation, injury and infection.
Objective This study aimed to estimate the incidence of vascular access-associated skin complications, and to identify patient, device, and healthcare-related characteristics associated with their development.
Method A secondary data analysis of 13 vascular access studies between 2008 and 2017 was undertaken, involving children and adults. A total of 7,669 participants with 10,859 devices were included, involving peripheral venous (n=9,933), peripheral arterial (n=341), and central venous access (n=585) devices.
Standardised study data were extracted into a single database. Cox proportional hazards regression models were used for time-to-event, per-device analyses, to examine risk factors. Univariate associations were undertaken, with p<0.01 significant.
Results Over 12% of devices were associated with skin complication, at 46.2 per 1000 catheter days for peripheral devices (95% confidence interval [CI] 42.1- 50.7), and 22.5 per 1000 catheter days for central devices (95% CI 16.5-306). The most common skin complications were bruising (peripheral n=134, 3.7%; central n= 33, 6.8%), infiltration for peripheral devices (n=296; 2.9%), and dermatitis for central devices (n=13; 2.2%). Risk factors were related to device (e.g., skin tears associated with peripheral arterial catheters [hazard ratio, HR 16.0], radial insertion [HR 18.0] basilic insertion [HR 26.0])) and patient characteristics (e.g., poor skin integrity associated with increased risk of peripheral device bruising [HR 4.12], infiltration [HR 1.98], and skin tear [HR 48.4]), rather than management approaches.
Conclusion Significant skin complications can develop surrounding vascular access devices, and these are associated with several modifiable and non-modifiable risk factors.
THE APHERESIS PORT FLOWS LIKE A DREAM: CASE STUDIES
Mrs Adrienne Woods1, Dr Juliana Teo1, Mrs Robyn Shoemark1
1The Children’s Hospital at Westmead, Westmead, Australia
Introduction Worldwide, automated red blood cell exchange (RBCX) transfusions are being used to successfully treat patients with sickle cell disease (SCD). At the Children’s Hospital at Westmead (CHW), we have had an apheresis RBCX program since May 2015, initially requiring two separate vascular access sites – usually both cubital fossas, which had significant impact on the quality of life for the patient while having the procedure.
Over time, with repeated venous access on a monthly basis, veins become more difficult to access. Often skilled nurses were not enough and anaesthetics were called upon to help with access, or to look for alternatives.
Cases Case 1 is a 9-year-old boy with an electively placed Vortex Port inserted in Sept 2015 for RBCX. This port was the only suitable port available of the time. With a straight needle emerging at a 90-degree angle to the patient from the port, dressing and securing the needle and lines are extremely cumbersome. The team were always on the lookout for an alternative that would be an improvement on the current design.
Since October 2018, a single needle RBCX program has been available reducing the need for dual venous access.
Case 2, December 2018, CHW was the first in Australia to use the Bard PowerFlow™ Implantable Apheresis IV Port. Two brothers underwent the procedure to have this port implanted with great success. Access, securing and dressing is easy. The design of the port allows for better flow rates making this our much needed improvement.
